Advertisements
Advertisements
प्रश्न
What is monosaccharide?
Advertisements
उत्तर
Monosaccharides are carbohydrates which do not hydrolyse further into smaller units of polyhydroxy aldehydes or ketones.
APPEARS IN
संबंधित प्रश्न
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Give two evidences for presence of formyl group in glucose.
Draw a neat diagram for the following:
Haworth formula of glucopyranose.
Glucose on oxidation with dilute nitric acid gives _______________
Write the name of the polysaccharide used for the commercial preparation of glucose.
Write the name of the unit to which glucose unit is linked to from sucrose.
Explain the preparation of glucose from sucrose.
Write chemical reaction for following conversions
glucose into glucoxime
Classify the following carbohydrates into monosaccharide, disaccharide, oligosaccharide and polysaccharide:
glucose, cellulose, maltose, stachyose
What is the action of the following reagents on glucose?
hydrogen iodide
Write a chemical reaction to convert glucose into glucose cyanohydrin.
Identify the bond that links the carbon of two adjacent monosaccharides.
Identify the given structure 'P' and 'Q'.

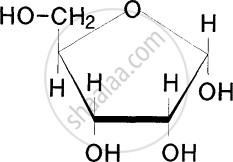
Identify the substances having glycosidic bond and peptide bond, respectively in their structure:
Which of the following is the basic unit of carbohydrates?
Carbohydrates can contain which of the following chemical groups?
The reserve food material in animals is ____________.
Identify the WRONG statement.
Which among the following compounds is obtained when glucose reacts with hydrogen cyanide?
Chitin is a/an ______.
The number of asymmetric carbon atoms in the glucose molecule is ____________.
Identify the CORRECT combination.
Which carbon atoms of α- D glucopyranose and β-D-fructofuranose respectively are linked together to form glycosidic linkage in sucrose?
Which among the following type of linkages is present in cellulose?
All these carbohydrates contain \[\ce{1 -> 4β}\] glycosidic linkage, EXCEPT ____________.
Which of the following monosaccharide is a ketohexose?
Which one of the following is a carbohydrate but does NOT follow the general formula of carbohydrate?
Which one of the following is Tetrose sugar?
Which of the following statements is NOT true for glyceraldehyde?
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
Which among the following statements is true for amylose?
Which one of the following sugar does NOT have same empirical fonnula as that of carbohydrate?
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Identify the number of oxygen atoms present in saccharic acid?
Which among the following reagents is used for conversion of glucose to glucoxime?
What is the molecular formula of glyceraldehyde?
Identify the product Y in the following reaction.
\[\ce{Maltose ->[Hydrolysis] X ->[dil. HNO3][(excess)] Y}\]
Which one of the following rotates the plane polarized light towards left?
α-D (+) Glucose and β-D (+) glucose are ____________.
What are reducing and non-reducing sugars?
Why carbohydrates are generally optically active?
Is the following sugar, D-sugar or L-sugar?

If 'n' represents total number of asymmetric carbon atoms in a compound, then the possible· number of optical isomers of the compound is ______.
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
Which one is a non-reducing commercial sugar?
Write Fischer projection formulae for glucose.
Write the ring structure of glucose.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Two monosaccharides are held together by ______ bond.
Which of the following is used to separate glucose and fructose from hydrolysate of sucrose?
