Topics
Laws of Motion
- Force and Motion
- Distance and Displacement
- Speed and Velocity
- Effect of Speed and Direction on Velocity
- Uniform and Non-uniform Motion
- Acceleration and Retardation
- Types of Acceleration
- Graphical Representation of Motion
- Displacement - Time Graph Or Distance - Time Graph
- Velocity - Time Graphs
- Equations of Motion by Graphical Method
- Derivation of Velocity - Time Relation by Graphical Method
- Derivation of Displacement - Time Relation by Graphical Method
- Derivation of Displacement - Velocity Relation by Graphical Method
- Uniform Circular Motion (UCM)
- Newton’s Laws of Motion
- Newton's First Law of Motion
- Newton’s Second Law of Motion
- Newton's Third Law of Motion
- Conservation of Linear Momentum and Its Principle
Work and Energy
Current Electricity
- Electricity
- Potential and Potential Difference
- Free Electrons
- Electric Current
- Ohm's Law
- Resistance and Resistivity of a Conductor
- Electric Circuit
- Symbols and Functions of Various Components of an Electric Circuits
- Electrical Conduction in Solids
- Experimental Verification of Ohm’s Law and Ohmic Resistors
- Resistance of a System of Resistors
- Resistors in Series
- Resistors in Parallel
- Domestic Electrical Connections
- Precautions to Be Taken While Using Electricity
Measurement of Matter
- Laws of Chemical Combination
- Law of Conservation of Matter (Law of Conservation of Mass)
- Law of Constant Proportions (Law of Definite Proportions)
- Atoms: Building Blocks of Matter
- Atomic Mass
- Symbols Used to Represent Atoms of Different Elements
- Molecules of Elements and Compounds
- Molecular Mass
- Mole Concept
- Avogadro's Number
- Periodic Trends in the Modern Periodic Table
- Variable Valency
- Ions (Radicals) and Its Types
- Chemical Formulae of Compounds
Acids, Bases and Salts
- Concept of Acid, Base, and Salt
- Ionic Compounds
- Dissociation of Ionic Compounds
- Arrhenius Theory of Acids and Bases
- Basicity and Acidity
- Ph of Solution
- Indicators
- Neutralization Reaction
- Reactions of Acids
- Reactions of Bases
- Salts
- Classification of Salts
- Water of Crystallization
- Electrolysis
- Electrolysis of Water
Classification of Plants
Energy Flow in an Ecosystem
Useful and Harmful Microbes
Environmental Management
- Weather and Climate
- Importance of Weather in the Living World
- Meteorology
- India Meteorological Department
- Solid Waste Management
- Waste and Its Categories
- Waste
- Harmful effects of solid waste
- Necessity of Solid Waste Management
- 7 Principles of Solid Waste Management
- Period Required for Degradation of Waste
- Disaster Management
- First Aid and Emergency Action
- Methods of Transporting Victims/Patients Safely
Information Communication Technology
- What is Information and Communication Technology ?
- Introduction to Computer
- Parts of Computer and Its Functions
- Architecture of Computer
- Computer Hardware and Software
- Microsoft Word
- Microsoft Excel
- Microsoft Powerpoint
- Applications of Technology in Science
- Opportunities in the field of computers
Reflection of Light
- Introduction to Light
- Mirrors
- Plane Mirror
- Spherical Mirrors
- Spherical Mirror > Concave Mirror
- Spherical Mirror > Convex Mirror
- Fundamental Terms Related to Spherical Mirrors
- Rules for Drawing Ray Diagrams
- Image Formation by Concave Mirror
- Image Formation by Convex Mirror
- Divergence and Convergence of Light
- Sign Convention
- Ray Optics - Mirror Formula
- Linear Magnification by Spherical Mirrors
Study of Sound
Carbon : An Important Element
- Carbon: A Versatile Element
- Straight chains, Branched chains, and Rings of Carbon atoms
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Non-crystalline/Amorphous Forms: Coal
- Non-crystalline/Amorphous Forms: Charcoal
- Hydrocarbons
- Non-crystalline/Amorphous Forms: Coke
- Solubility of Carbon
- Reaction of Carbon
- Carbon Dioxide
- Fire Extinguisher
- Methane
- Biogas Plant
Substances in Common Use
- Important Salts in Daily Life
- Properties and Uses of Sodium Chloride
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Some Crystalline Salts
- Soap
- Radioactivity
- Nature of Radioactive Radiation
- Characteristics of Alpha, Beta and Gamma Rays
- Uses of Radioactive Isotopes
- Harmful Effects of Radiation
- Chemical Substances in Day to Day Life
- Food Colours and Essences
- Dye
- Artificial Colours
- Deodorant
- Teflon
- Powder Coating
- Anodizing
- Ceramic
Life Processes in Living Organisms
- Transportation in Living Organisms
- Transportation in Plants
- Transportation of Water
- Transportation of Food and Other Substances
- Excretion
- Excretion in Plants
- Human Excretory System
- Dialysis and Artificial Kidney
- Need for Control and Coordination in Organisms
- Control and Co-ordination in Plants
- Control and Co-ordination in Human Being
- Nervous Control
- Human Nervous System
- Central Nervous System (CNS)
- Chemical Control
- Endocrine Glands: Location and Important Functions
Heredity and Variation
- Inherited Traits and Expression of Traits
- Chromosomes - The Carriers of Heredity
- Types of Chromosomes
- Deoxyribonucleic Acid (DNA)
- Ribonucleic acid (RNA)
- Gregor Johann Mendel – Father of Genetics
- Monohybrid Cross
- Dihybrid Cross
- Human Genetic Disorders
- Diseases Occuring Due to Mutation in Single Gene (Monogenic Disorders)
- Mitochondrial Disorder
- Disorders Due to Mutations in Multiple Genes : (Polygenic Disorders)
Introduction to Biotechnology
- Tissues - “The Teams of Workers”
- Animal Tissues
- Epithelial Tissue
- Connective Tissue
- Muscular Tissue
- Nervous Tissue
- Plant Tissues
- Meristems or Meristematic Tissues
- Permanent Tissue
- Simple Permanent Tissues (Supporting Tissue)
- Complex Permanent Tissues
- Tissue Culture
- Changes in Agricultural Management Due to Biotechnology
- Application of Biotechnology in Floriculture, Nurseries and Forestry
- Agritourism
- Animal Husbandry (Livestock)
- Animal Husbandry (Livestock) > Poultry Farm Management
- Animal Husbandry (Livestock) > Sericulture
Observing Space : Telescopes
- Introduction
- Experiment
Introduction:
Electrolysis of water is a fundamental chemical process used to decompose water (H₂O) into its constituent gases, hydrogen (H₂) and oxygen (O₂), by passing an electric current through it. This process requires an electrolyte to increase the conductivity of pure water, which is otherwise a poor conductor of electricity. Common electrolytes like salt (NaCl), dilute sulphuric acid (H₂SO₄), or sodium hydroxide (NaOH) are added for effective electrolysis.
- The process takes place in an electrolytic cell where two electrodes (cathode and anode) are submerged in an electrolyte solution.
- When a direct current (DC) is applied, water molecules dissociate: hydrogen gas is released at the cathode (negative electrode), and oxygen gas is released at the anode (positive electrode).
- The volume of hydrogen gas formed is approximately twice that of oxygen, consistent with the molecular composition of water (H₂O).
- Electrolysis of water is not only a practical demonstration of water's composition but also a significant industrial process.
- It is widely used for producing hydrogen gas, which serves as a clean energy carrier and is critical for applications like fuel cells, ammonia production, and refining metals.
Experiment
1. Aim: To demonstrate the electrolysis of water and observe the formation of hydrogen and oxygen gases at the cathode and anode, respectively.
2. Requirements: 500 ml of pure water, 2 g of salt, a beaker (500 ml capacity), test tubes (2), electrical wires with cladding removed (2 cm at the ends), DC power supply (6V potential difference), litmus paper, and optional electrolytes: dilute H₂SO₄ and dilute NaOH.
3. Procedure
- Dissolve 2 g of salt in 500 ml of pure water and take 250 ml of the solution in a beaker.
- Connect two wires to the terminals of a 6V power supply and immerse their exposed ends as electrodes in the solution.
- Fill two test tubes with the solution and invert them over the electrodes without trapping air.
- Turn on the power supply, allowing current to pass through, and observe gas bubbles forming in the test tubes.
- Note the difference in gas volumes; test the solutions with litmus paper to identify acidity and alkalinity.
- Repeat using dilute H₂SO₄ and dilute NaOH as electrolytes and record observations.
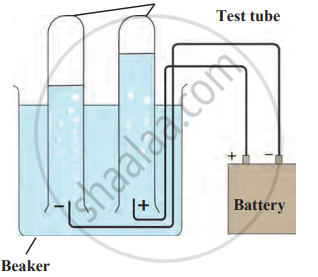
Electrolysis of water
4. Observations: Gas bubbles form near both electrodes. The volume of gas collected at the cathode is approximately double that of the gas collected at the anode. Testing with litmus paper shows the solution near the cathode is basic (turns blue) and near the anode is acidic (turns red).
5. Conclusion: The experiment shows the electrolysis of water-
- Hydrogen gas forms at the cathode (2H₂O + 2e⁻ → H₂ + 2OH⁻).
- Oxygen gas forms at the anode (2H₂O → O₂ + 4H⁺ + 4e⁻).
The volume ratio of hydrogen to oxygen is 2:1, confirming the composition of water (H₂O). Using dilute H₂SO₄ or NaOH enhances conductivity and results in similar observations.
