Topics
Some Basic Concepts of Chemistry
Introduction to Analytical Chemistry
- Introduction of Analytical Chemistry
- Analysis
- Mathematical Operation and Error Analysis
- Determination of Molecular Formula
- Chemical Reactions and Stoichiometric Calculations
- Limiting Reagent
- Concentration of a Solution
- Use of Graph in Analysis
Basic Analytical Techniques
- Introduction of Some Analytical Techniques
- Purification of Solids
- Crystallisation Method
- Fractional Crystallization
- Simple Distillation Method
- Solvent Extraction
- Chromatography Method
- Chromatography Method > Adsorption Chromatography
- Chromatography Method > Partition Chromatography
Structure of Atom
Chemical Bonding
- Concept of Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel and Lewis Approach to Chemical Bonding
- Formal Charge
- Limitations of the Octet Rule
- Valence Shell Electron Pair Repulsion (VSEPR) Theory
- Valence Bond Theory (VBT)
- Molecular Orbital Theory
- Parameters of Covalent Bond
- Dipole Moment
- Resonance
Redox Reactions
Modern Periodic Table
- Introduction of Periodic Table
- Structure of the Modern Periodic Table
- Periodic Table and Electronic Configuration
- Blockwise Characteristics of Elements
- Periodic Trends in Elemental Properties
Elements of Group 1 and 2
Elements of Group 13, 14 and 15
- Electronic Configuration of Elements of Groups 13, 14 and 15
- Trends in Atomic and Physical Properties of Elements of Groups 13, 14 and 15
- Chemical Properties of the Elements of the Groups 13,14 and 15
- Carbon: A Versatile Element
- Allotropes of Carbon > Diamond
- Molecular Structures of Some Important Compounds of the Group 13, 14 and 15 Elements
- Chemistry of Notable Compounds of Elements of Groups 13, 14 and 15
States of Matter
Adsorption and Colloids
Chemical Equilibrium
- Introduction of Chemical Equilibrium
- Equilibrium in Physical Processes
- Equilibrium in Chemical Processes - Dynamic Equilibrium
- Law of Mass Action and Equilibrium Constant
- Homogeneous Equilibria
- Characteristics of Equilibrium Constant
- Applications of Equilibrium Constants
- Le Chaterlier's Principle and Factors Altering the Composition of Equilibrium
- Industrial Application
Nuclear Chemistry and Radioactivity
- Introduction: Nuclear Chemistry is a Branch of Physical Chemistry
- Classification of Nuclides
- Nuclear Stability
- Radioactivity
- Radioactive Decays
- Modes of Decay
- Nuclear Reactions
- Applications of Radio Isotopes
Basic Principles of Organic Chemistry
- Organic Chemistry
- Structural Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- Isomerism
- Theoretical Basis of Organic Reactions
Hydrocarbons
Chemistry in Everyday Life
Three States of Matter
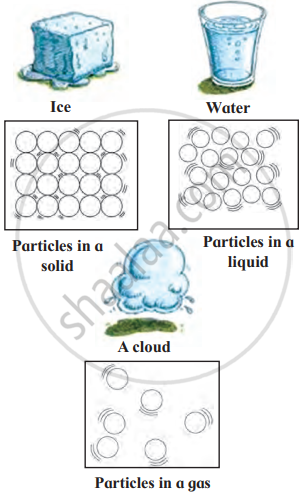
1. The Solid State: Solids have a definite shape and fixed size. The particles in solids are packed tightly together, which is why solids are usually hard and firm. Solids cannot be easily compressed or squashed because there is no space between the particles.
Example of Solids:
- Ice is a solid form of water, and it has a definite shape.
- Other examples include wood, stone, pencils, and books.
2. The Liquid State: Liquids do not have a definite shape, but they do have a fixed volume. The particles in a liquid are more loosely arranged than in a solid, which allows liquids to flow and take the shape of the container they are in. Liquids are not easily compressed, as the particles are still fairly close together.
Example of Liquids:
- Water is a liquid. It flows and takes the shape of any container, like a glass or bottle.
- Other examples of liquids include milk, juice, and oil.
3. The Gaseous State: Gases do not have a definite shape or a fixed volume. They will spread out and fill any space they are in. The particles in gases are very far apart and move around freely in all directions. Gases can be easily compressed because there is a lot of space between the particles.
Example of Gases:
- Air is a mixture of gases, including oxygen and nitrogen. You cannot see air, but it fills the space around us.
- Other examples include steam, carbon dioxide, and helium (which is used in balloons).
Maharashtra State Board: Class 12
Key Points: States of Matter
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Mean molecular separation | ~3–5 Å | ~3–10 Å | >5 Å |
| Particle arrangement | Tightly packed, regular | Loosely packed, irregular | Highly irregular |
| Particle movement | Fixed positions, cannot move freely | Moves a small distance within liquid | Continuous random motion |
| Shape & volume | Definite shape and volume | Takes shape of container, definite volume | Takes shape and volume of container |
| Intermolecular space | Very small | Moderate | Large |
| Effect of temperature | Small volume change | Moderate volume change | Significant volume change |
| Compressibility | Practically incompressible | Slightly compressible | Highly compressible |
| Example | A piece of iron | Water, spirit, oil | Air |
