Please select a subject first
Advertisements
Advertisements
The complex [Co(NH3)5(NO2)]Cl2 is red in colour. Give IUPAC the name of its linkage isomer.
Concept: Isomerism in Coordination Compounds >> Structural Isomerism
Name the method used for the refining of Nickel metal.
Concept: Refining of Crude Metals
Which would undergo SN1 reaction faster in the following pair and why?

Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
In the extraction of Al, impure Al2O3 is dissolved in conc. NaOH to form sodium aluminate and leaving impurities behind. What is the name of this process?
Concept: Occurrence of Metals
Which would undergo SN2 reaction faster in the following pair and why ?

Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Indicate the principle behind the method used for the refining of zinc.
Concept: Occurrence of Metals
Which form of the iron is the purest form of commercial iron?
Concept: Occurrence of Metals
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Concept: Physical Properties of Haloalkanes and Haloarenes
The presence of nitro group (−NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
Concept: Reactions of Haloarenes - Nucleophilic Substitution
What is the principle behind the zone refining of metals?
Concept: Refining of Crude Metals
Explain the principle of the method of electrolytic refining of metals. Give one example.
Concept: Refining of Crude Metals
Write the structures of A, B and C in the following:

Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Concept: Physical Properties of Haloalkanes and Haloarenes
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Concept: Reactions of Haloarenes - Nucleophilic Substitution
Which reducing agent is employed to get copper from the leached low-grade copper ore?
Concept: Types of Separation or Concentration of an Ore >> Leaching
Draw the structure of major monohalo product in each of the following reactions :
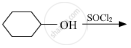
Concept: Classification of Haloalkanes and Haloarenes
Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
\backslash\phantom{...........}\\
\ce{CH - CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{..................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Concept: Physical Properties of Haloalkanes and Haloarenes
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Concept: Physical Properties of Haloalkanes and Haloarenes
