Please select a subject first
Advertisements
Advertisements
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Complete the reaction with the main product formed:

Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Convert bromoethane to propanamine.
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
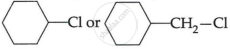
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
Concept: Reactions of Haloalkanes - Nucleophilic Substitution Reactions
Account for the following:
Helium is used in diving apparatus.
Concept: Group 18 Elements - The Noble gas Family
Draw the structures of the following molecules: XeF2
Concept: Group 18 Elements - The Noble gas Family
Draw the structures of the following: H4P2O7 (Pyrophosphoric acid)
Concept: Group 15 Elements - The Nitrogen Family >> Oxoacids of Phosphorus
What happens when
H3PO3 is heated ?
Write the reactions involved.
Concept: Group 15 Elements - The Nitrogen Family >> Phosphine
Write the mechanism of the following reaction:

Concept: Chemical Reactions of Alcohols and Phenols >> Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols
Write the main product(s) in each of the following reactions:

Concept: Chemical Reaction of Ethers - Cleavege of C-O Bonds
Write the main product(s) in each of the following reactions:

Concept: Methods of Preparation of Alcohols
Account for the following :
Ozone is thermodynamically unstable.
Concept: Group 16 Elements - The Oxygen Family >> Ozone
Account for the following : Solid PCl5 is ionic in nature.
Concept: Group 15 Elements - The Nitrogen Family >> Phosphorus Halides
Account for the following:
Fluorine forms only one oxoacid HOF.
Concept: Group 17 Elements - The Halogen Family >> Oxoacids of Halogens
Draw the structure of BrF5
Concept: Group 17 Elements - The Halogen Family >> Interhalogen Compounds
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Concept: Group 17 Elements - The Halogen Family
Write the conditions to maximize the yield of H2SO4 by contact process.
Concept: Group 16 Elements - The Oxygen Family >> Sulphuric Acid
What is the basicity of H3PO4?
Concept: Group 15 Elements - The Nitrogen Family >> Oxoacids of Phosphorus
Name the reagents used in the following reactions:

Concept: Methods of Preparation of Alcohols
