Advertisements
Advertisements
Question
Which of the following diagrams (Figure) depicts ideal gas behaviour?
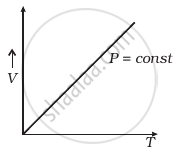 (a) |
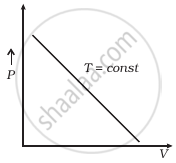 (b) |
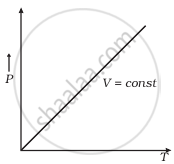 (c) |
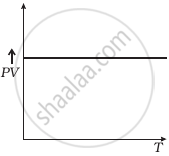 (d) |
Advertisements
Solution
a and c
Explanation:
For ideal gas behaviour,
PV = nRT ......(i)
(a) When pressure, P = constant
From (i) Volume V ∝ Temperature T
The graph of V versus T will be a straight line.
(b) When T = constant
From (i) PV = constant
So, the graph of P versus V will be a rectangular hyperbola. Hence this graph is wrong. The correct graph is shown below:
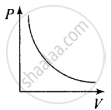
(c) When V = constant
From (i) P ∝ T
So, the graph is a straight line passing through the origin.
(d) From (i) PV ∝ T
⇒ `(PV)/T` = constant
So, the graph of PV versus T will be a straight line parallel to the temperature axis (x-axis).
i.e., the slope of this graph will be zero.
So, (d) is not correct.
APPEARS IN
RELATED QUESTIONS
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
Estimate the total number of air molecules (inclusive of oxygen, nitrogen, water vapour and other constituents) in a room of capacity 25.0 m3 at a temperature of 27 °C and 1 atm pressure
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C? (atomic mass of Ar = 39.9 u, of He = 4.0 u).
What do you understand by gas?
What is diffusion? Give an example to illustrate it.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
Correct the following statement:
0°C is equal to zero Kelvin.
Name or state the following:
The absolute temperature value corresponding to 35°C.
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
The average energy per molecule is proportional to ______
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Show that for diatomic gas the ratio of the two specific heats is 7:5.
Gases exert pressure on the walls of the container because the gas molecules ______
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
