Advertisements
Advertisements
Question
Name or state the following:
The standard pressure of a gas in cm. of mercury corresponding to one atmospheric pressure.
Advertisements
Solution
76 cm.
APPEARS IN
RELATED QUESTIONS
Oxygen is filled in a closed metal jar of volume 1.0 × 10−3 m3 at a pressure of 1.5 × 105Pa and temperature 400 K. The jar has a small leak in it. The atmospheric pressure is 1.0 × 105 Pa and the atmospheric temperature is 300 K. Find the mass of the gas that leaks out by the time the pressure and the temperature inside the jar equalise with the surrounding.
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
During the practical session in the lab when hydrogen sulphide gas having offensive odour is prepared for some test, we can smell the gas even 50 metres away. Explain the phenomenon.
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
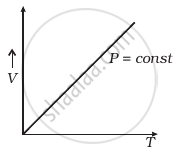 (a) |
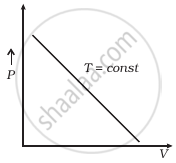 (b) |
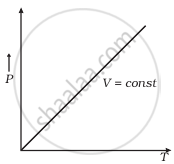 (c) |
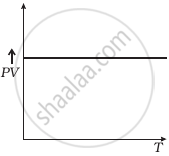 (d) |
Calculate the number of atoms in 39.4 g gold. Molar mass of gold is 197g mole–1.
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
