Advertisements
Advertisements
Question
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Advertisements
Solution
According to Boyle's Law V α `1/"p"`
V = K.`1/"p"`
∴ P × V = K = a constant
∴ Product of volume and pressure of a given mass of dry gas is constant. [at constant temperature]
APPEARS IN
RELATED QUESTIONS
An oxygen cylinder of volume 30 litres has an initial gauge pressure of 15 atm and a temperature of 27 °C. After some oxygen is withdrawn from the cylinder, the gauge pressure drops to 11 atm and its temperature drops to 17 °C. Estimate the mass of oxygen taken out of the cylinder (R = 8.31 J mol–1 K–1, molecular mass of O2 = 32 u)
Estimate the average thermal energy of a helium atom at the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
Choose the correct answer:
The graph of PV vs P for gas is
Fill in the blanks:
If the temperature is reduced to half, ………….. would also reduce to half.
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
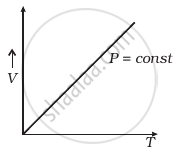 (a) |
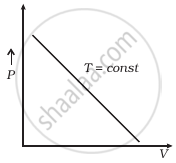 (b) |
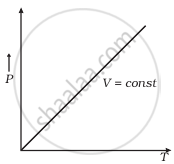 (c) |
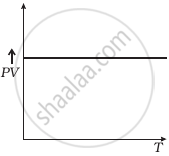 (d) |
