Advertisements
Advertisements
Question
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Advertisements
Solution
The interparticle attraction is weak and interparticle space is large in gases because the particles are completely free to move randomly in the entire available space and take the shape of the vessel in which the gas is kept.
APPEARS IN
RELATED QUESTIONS
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
Oxygen is filled in a closed metal jar of volume 1.0 × 10−3 m3 at a pressure of 1.5 × 105Pa and temperature 400 K. The jar has a small leak in it. The atmospheric pressure is 1.0 × 105 Pa and the atmospheric temperature is 300 K. Find the mass of the gas that leaks out by the time the pressure and the temperature inside the jar equalise with the surrounding.
50 m3 of saturated vapour is cooled down from 30°C to 20°C. Find the mass of the water condensed. The absolute humidity of saturated water vapour is 30 g m−3 at 30°C and 16 g m−3 at 20°C.
Name or state the following:
The standard pressure of a gas in cm. of mercury corresponding to one atmospheric pressure.
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Which of the following diagrams (Figure) depicts ideal gas behaviour?
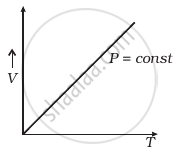 (a) |
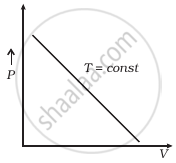 (b) |
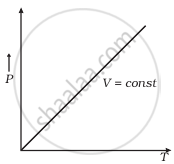 (c) |
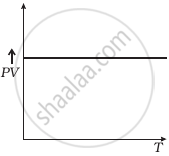 (d) |
A box of 1.00 m3 is filled with nitrogen at 1.50 atm at 300K. The box has a hole of an area 0.010 mm2. How much time is required for the pressure to reduce by 0.10 atm, if the pressure outside is 1 atm.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
P ∝ T at constant volume is the statement of ______.
