Advertisements
Advertisements
Question
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Advertisements
Solution
Gas equation.
APPEARS IN
RELATED QUESTIONS
Estimate the average thermal energy of a helium atom at the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
At what temperature is the root mean square speed of an atom in an argon gas cylinder equal to the rms speed of a helium gas atom at – 20 °C? (atomic mass of Ar = 39.9 u, of He = 4.0 u).
During the practical session in the lab when hydrogen sulphide gas having offensive odour is prepared for some test, we can smell the gas even 50 metres away. Explain the phenomenon.
What is diffusion? Give an example to illustrate it.
Choose the correct answer:
The graph of PV vs P for gas is
Name or state the following:
The absolute temperature value corresponding to 35°C.
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Which of the following diagrams (Figure) depicts ideal gas behaviour?
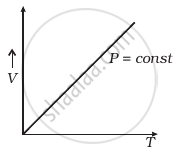 (a) |
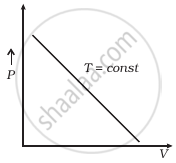 (b) |
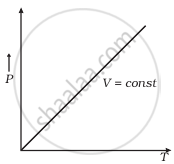 (c) |
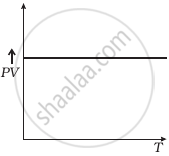 (d) |
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
