Advertisements
Advertisements
प्रश्न
Which of the following diagrams (Figure) depicts ideal gas behaviour?
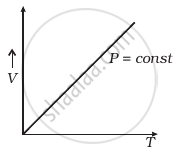 (a) |
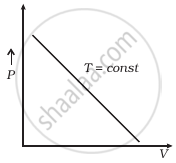 (b) |
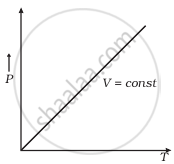 (c) |
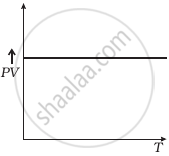 (d) |
Advertisements
उत्तर
a and c
Explanation:
For ideal gas behaviour,
PV = nRT ......(i)
(a) When pressure, P = constant
From (i) Volume V ∝ Temperature T
The graph of V versus T will be a straight line.
(b) When T = constant
From (i) PV = constant
So, the graph of P versus V will be a rectangular hyperbola. Hence this graph is wrong. The correct graph is shown below:
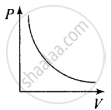
(c) When V = constant
From (i) P ∝ T
So, the graph is a straight line passing through the origin.
(d) From (i) PV ∝ T
⇒ `(PV)/T` = constant
So, the graph of PV versus T will be a straight line parallel to the temperature axis (x-axis).
i.e., the slope of this graph will be zero.
So, (d) is not correct.
APPEARS IN
संबंधित प्रश्न
Estimate the average thermal energy of a helium atom at the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
Oxygen is filled in a closed metal jar of volume 1.0 × 10−3 m3 at a pressure of 1.5 × 105Pa and temperature 400 K. The jar has a small leak in it. The atmospheric pressure is 1.0 × 105 Pa and the atmospheric temperature is 300 K. Find the mass of the gas that leaks out by the time the pressure and the temperature inside the jar equalise with the surrounding.
What do you understand by gas?
What is diffusion? Give an example to illustrate it.
Choose the correct answer:
The graph of PV vs P for gas is
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Name or state the following:
The absolute temperature value corresponding to 35°C.
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Give reason for the following:
Volumes of gases are converted into s.t.p. conditions and then compared.
The average energy per molecule is proportional to ______
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Show that for diatomic gas the ratio of the two specific heats is 7:5.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
The volume V of an enclosure contains a mixture of three gases, 16 g of oxygen, 28 g of nitrogen and 44 g of carbon dioxide at absolute temperature T. Consider R as universal gas constant. The pressure of the mixture of gases is ______.
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
