Advertisements
Advertisements
प्रश्न
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Advertisements
उत्तर
At room temperature, T = 27°C = 300 K
Average thermal energy = `3/2` kT
Where k is Boltzmann constant = 1.38 × 10–23 m2 kg s–2 K–1
`therefore 3/2kT = 3/2 xx 1.38 xx 10^(-38) xx 300`
= 6.21 × 10–21J
Hence, the average thermal energy of a helium atom at room temperature (27°C) is 6.21 × 10–21 J.
संबंधित प्रश्न
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP: 1 atmospheric pressure, 0 °C). Show that it is 22.4 litres
During the practical session in the lab when hydrogen sulphide gas having offensive odour is prepared for some test, we can smell the gas even 50 metres away. Explain the phenomenon.
What is diffusion? Give an example to illustrate it.
Give reasons for the following:
Gas fills the vessel completely in which it is kept.
Choose the correct answer:
The graph of PV vs P for gas is
Match the following:
|
|
Column A |
Column B |
|
(a) |
cm3 |
(i) Pressure |
|
(b) |
Kelvin |
(ii) Temperature |
|
(c) |
Torr |
(iii) Volume |
|
(d) |
Boyle's law |
(iv) `"V"/"T" = ("V"_1)/("T"_1)` |
|
(a) |
Charles's law |
(v) `"PV"/"T" = ("P"_1 "V"_1)/"T"_1` |
|
|
|
(vi) PV = P1V1 |
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Correct the following statement:
0°C is equal to zero Kelvin.
Name or state the following:
The absolute temperature value corresponding to 35°C.
The average energy per molecule is proportional to ______
If the absolute temperature of a body is doubled, the power radiated will increase by a factor of ______
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Show that for diatomic gas the ratio of the two specific heats is 7:5.
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
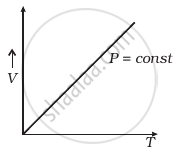 (a) |
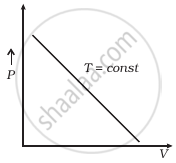 (b) |
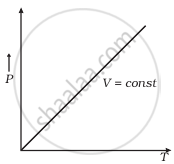 (c) |
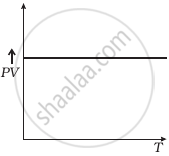 (d) |
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
