Advertisements
Advertisements
Question
Ionisation enthalpy (∆iH1kJ mol–1) for the elements of Group 13 follows the order.
Options
B > Al > Ga > In > Tl
B < Al < Ga < In < Tl
B < Al > Ga < In > Tl
B > Al < Ga > In < Tl
Advertisements
Solution
B > Al < Ga > In < Tl
Explanation:
On moving down the group from B to Tl, a regular decreasing trend in the ionisation energy values is not observed.
| Element | B | Al | Ga | In | Tl |
| I.E. (kJ/mol) | 801 | 577 | 579 | 558 | 589 |
In Ga, there are ten d-electrons in the penultimate shell which screen the nuclear charge less effectively and thus, outer electron is held firmly. As a result, the ionisation energy of both A1 and Ga is nearly the same. The increase in ionisation energy from In to Tl is due to poor screening effect of 14f electrons present in the inner shell.
APPEARS IN
RELATED QUESTIONS
If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
What happens when BF3 is reacted with ammonia?
In the structure of diborane ______.
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
The most commonly used reducing agent is ______.
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Which of the following statements are correct. Answer on the basis of Figure.
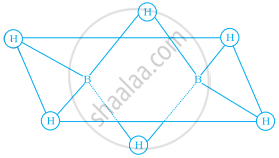
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]– only whereas AlCl3 in acidified aqueous solution forms [Al(H2O)6]3+ ion. Explain what is the hybridisation of boron and aluminium in these species?
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Explain the following:
PbX2 is more stable than PbX4.
Explain the following:
Tl (NO3)3 acts as an oxidising agent.
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Ionisation enthalpy
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Nature of halides
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Boron compounds behave as Lewis acids because of their ______.
