Advertisements
Advertisements
Question
If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
Advertisements
Solution 1
As a result of the difference in the electronegativities of B and Cl, the B–Cl bond is polar in nature. However, the BCl3 molecule is non-polar. This is because BCl3 is trigonal planar in shape. It is a symmetrical molecule. Hence, the respective dipole-moments of the B–Cl bond cancel each other, thereby causing a zero-dipole moment.

Solution 2
B-Cl bond has dipole moment because of polarity. In BCl3 since the molecule is symmetrical (planar). Thus the polarities cancel out.
RELATED QUESTIONS
Suggest reasons why the B–F bond lengths in BF3 (130 pm) and `"BF"_4^(-)` (143 pm) differ.
How would you explain the lower atomic radius of Ga as compared to Al?
What do you understand by inert pair effect?
Write a balanced equation for Al + NaOH → ?
Which of the following oxides is acidic in nature?
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
The most commonly used reducing agent is ______.
Dry ice is ______.
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Which of the following statements are correct. Answer on the basis of Figure.
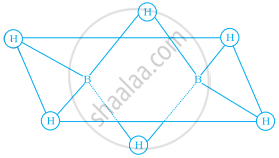
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
BCl3
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Explain the following:
Tl (NO3)3 acts as an oxidising agent.
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Match the species given in Column I with the properties mentioned in Column II.
| Column I | Column II |
| (i) \[\ce{BF^{-}4}\] | (a) Oxidation state of central atom is +4 |
| (ii) AICI3 | (b) Strong oxidising agent |
| (iii) SnO | (c) Lewis acid |
| (iv) PbO2 | (d) Can be further oxidised |
| (e) Tetrahedral shape |
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
