Advertisements
Advertisements
Question
Which of the following statements are correct. Answer on the basis of Figure.
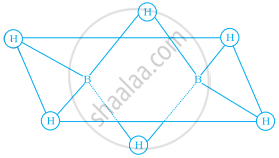
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Advertisements
Solution
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explanation:
Each of the two boron atoms is in sp3 – hybrid state. Of the four hybrid orbitals, three have one electron each while the fourth is empty. Two of the four orbitals of each, of the boron atom overlap with two terminal hydrogen atoms forming two normal B – H σ-bonds. One of the remaining hybrid orbitals (either empty or singly occupied) of one of the boron atoms, 15-orbital of H (bridge atom) and one of hybrid orbitals of the other boron atom overlap to form a delocalized orbital covering the three nuclei with a pair of electrons. This is three-centre two-electron bond. Similar overlapping occurs with the second hydrogen atom (bridging) forming three centre two electrons bond.
APPEARS IN
RELATED QUESTIONS
How can you explain higher stability of BCl3 as compared to TlCl3?
Suggest reasons why the B–F bond lengths in BF3 (130 pm) and `"BF"_4^(-)` (143 pm) differ.
What happens when BF3 is reacted with ammonia?
Write a balanced equation for Al + NaOH → ?
Ionisation enthalpy (∆iH1kJ mol–1) for the elements of Group 13 follows the order.
The most commonly used reducing agent is ______.
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Explain why the following compounds behave as Lewis acids?
BCl3
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Explain the following:
Boron does not exist as B3+ ion.
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Complete the following chemical equations:
\[\ce{Z + 3 LiAlH4 -> X + 3LiF + 3AlF_3}\]
\[\ce{X + 6H2 -> Y + 6H2}\]
\[\ce{3X + 3O2 ->[Δ] B2O3 + 3H2O}\]
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Ionisation enthalpy
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Boron fluoride exists as BF3 but boron hydride doesn’t exist as BH3. Give reason. In which form does it exist? Explain its structure.
A group 13 element ‘X’ reacts with chlorine gas to produce a compound XCl3. XCl3 is electron deficient and easily reacts with NH3 to form \[\ce{Cl3X –> NH3}\] adduct; however, XCl3 does not dimerize X is ______.
