Advertisements
Advertisements
Question
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Advertisements
Solution
As we move down the group in group 13 the participation of s-electrons in bond formation decreases the primary reason behind this is the inert pair effect. In this the p-electrons take part in bond formation and more energy is required to unpair the valence electrons to make them participate in bonding. Due to this the lower oxidation state of elements becomes stable than the higher oxidation state. As for thallium, +1 oxidation state is more stable than +3.
APPEARS IN
RELATED QUESTIONS
If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
What do you understand by inert pair effect?
Write a balanced equation for Al + NaOH → ?
Write a balanced equation for B2H6 + NH3 → ?
Which of the following oxides is acidic in nature?
In the structure of diborane ______.
Dry ice is ______.
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Which of the following statements are correct. Answer on the basis of Figure.
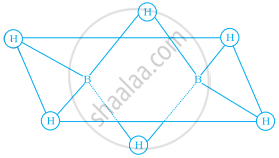
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
BCl3
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Match the species given in Column I with the properties mentioned in Column II.
| Column I | Column II |
| (i) \[\ce{BF^{-}4}\] | (a) Oxidation state of central atom is +4 |
| (ii) AICI3 | (b) Strong oxidising agent |
| (iii) SnO | (c) Lewis acid |
| (iv) PbO2 | (d) Can be further oxidised |
| (e) Tetrahedral shape |
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Oxidation states
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
Which one of the following is the correct statement?
