Advertisements
Advertisements
Question
Which of the following oxides is acidic in nature?
Options
B2O3
Al2O3
Ga2O3
In2O3
Advertisements
Solution
B2O3
Explanation:
B2O3 is acidic in nature. It reacts with basic oxides to form metal borates. Acidic nature decreases on moving down the group.
APPEARS IN
RELATED QUESTIONS
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
What do you understand by inert pair effect?
Write a balanced equation for Al + NaOH → ?
Write a balanced equation for B2H6 + NH3 → ?
The geometry of a complex species can be understood from the knowledge of type of hybridisation of orbitals of central atom. The hybridisation of orbitals of central atom in [Be(OH)4]– and the geometry of the complex are respectively.
Ionisation enthalpy (∆iH1kJ mol–1) for the elements of Group 13 follows the order.
In the structure of diborane ______.
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
Which of the following statements are correct. Answer on the basis of Figure.
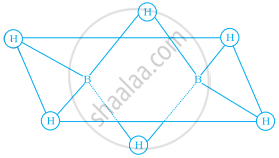
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain the following:
Boron does not exist as B3+ ion.
Explain the following:
PbX2 is more stable than PbX4.
Explain the following:
Tl (NO3)3 acts as an oxidising agent.
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Complete the following chemical equations:
\[\ce{Z + 3 LiAlH4 -> X + 3LiF + 3AlF_3}\]
\[\ce{X + 6H2 -> Y + 6H2}\]
\[\ce{3X + 3O2 ->[Δ] B2O3 + 3H2O}\]
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Ionisation enthalpy
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Nature of halides
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
