Advertisements
Advertisements
प्रश्न
Which of the following oxides is acidic in nature?
पर्याय
B2O3
Al2O3
Ga2O3
In2O3
Advertisements
उत्तर
B2O3
Explanation:
B2O3 is acidic in nature. It reacts with basic oxides to form metal borates. Acidic nature decreases on moving down the group.
APPEARS IN
संबंधित प्रश्न
Suggest reasons why the B–F bond lengths in BF3 (130 pm) and `"BF"_4^(-)` (143 pm) differ.
What happens when BF3 is reacted with ammonia?
Write a balanced equation for B2H6 + NH3 → ?
The exhibition of highest co-ordination number depends on the availability of vacant orbitals in the central atom. Which of the following elements is not likely to act as central atom in \[\ce{MF^{3-}6}\]?
In the structure of diborane ______.
Dry ice is ______.
Which of the following statements are correct. Answer on the basis of Figure.
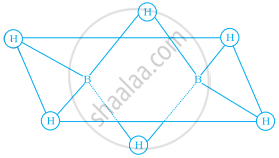
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
AlCl3
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]– only whereas AlCl3 in acidified aqueous solution forms [Al(H2O)6]3+ ion. Explain what is the hybridisation of boron and aluminium in these species?
Explain the following:
PbX2 is more stable than PbX4.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Match the species given in Column I with the hybridisation given in Column II.
| Column I | Column II |
| (i) Boron in [B(OH)4]– | (a) sp2 |
| (ii) Aluminium in [Al(H2O)6]3+ | (b) sp3 |
| (iii) Boron in B2H6 | (c) sp3d2 |
| (iv) Carbon in Buckminsterfullerene | |
| (v) Silicon in \[\ce{SiO^{4-}4}\] | |
| (vi) Germanium in [GeCl6]2– |
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Ionisation enthalpy
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
TlCl3, TlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
