Advertisements
Advertisements
प्रश्न
In the structure of diborane ______.
पर्याय
All hydrogen atoms lie in one plane and boron atoms lie in a plane perpendicular to this plane.
2 boron atoms and 4 terminal hydrogen atoms lie in the same plane and 2 bridging hydrogen atoms lie in the perpendicular plane.
4 bridging hydrogen atoms and boron atoms lie in one plane and two terminal hydrogen atoms lie in a plane perpendicular to this plane.
All the atoms are in the same plane.
Advertisements
उत्तर
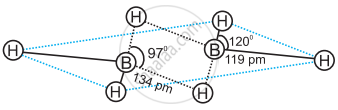
In the structure of diborane 2 boron atoms and 4 terminal hydrogen atoms lie in the same plane and 2 bridging hydrogen atoms lie in the perpendicular plane.
Explanation:
Four terminal hydrogen atoms and two boron atoms lie in the same plane and two hydrogen atoms forming bridges lie in a plane perpendicular to the rest of the molecule.
APPEARS IN
संबंधित प्रश्न
How can you explain higher stability of BCl3 as compared to TlCl3?
What happens when BF3 is reacted with ammonia?
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Write a balanced equation for B2H6 + NH3 → ?
Dry ice is ______.
Which of the following statements are correct. Answer on the basis of Figure.
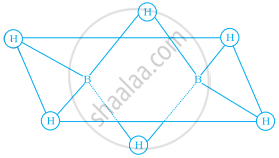
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain why the following compounds behave as Lewis acids?
AlCl3
Explain the following:
Boron does not exist as B3+ ion.
Explain the following:
Pb4+ acts as an oxidising agent but Sn2+ acts as a reducing agent.
Identify the compounds A, X and Z in the following reactions:
\[\ce{A + 2HCl + 5H2O -> 2NaCl + X}\]
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Complete the following chemical equations:
\[\ce{Z + 3 LiAlH4 -> X + 3LiF + 3AlF_3}\]
\[\ce{X + 6H2 -> Y + 6H2}\]
\[\ce{3X + 3O2 ->[Δ] B2O3 + 3H2O}\]
Match the species given in Column I with the properties mentioned in Column II.
| Column I | Column II |
| (i) \[\ce{BF^{-}4}\] | (a) Oxidation state of central atom is +4 |
| (ii) AICI3 | (b) Strong oxidising agent |
| (iii) SnO | (c) Lewis acid |
| (iv) PbO2 | (d) Can be further oxidised |
| (e) Tetrahedral shape |
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Atomic size
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Metallic character
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
AlCl3 , AlCl
A nonmetallic element of group 13, used in making bullet proof vests is extremely hard solid of black colour. It can exist in many allotropic forms and has unusually high melting point. Its trifluoride acts as Lewis acid towards ammonia. The element exihibits maximum covalency of four. Identify the element and write the reaction of its trifluoride with ammonia. Explain why does the trifluoride act as a Lewis acid.
Boron compounds behave as Lewis acids because of their ______.
Which one of the following is the correct statement?
