Advertisements
Advertisements
प्रश्न
How can you explain higher stability of BCl3 as compared to TlCl3?
Advertisements
उत्तर
Boron and thallium belong to group 13 of the periodic table. In this group, the +1 oxidation state becomes more stable on moving down the group. BCl3 is more stable than TlCl3 because the +3 oxidation state of B is more stable than the +3 oxidation state of Tl. In Tl, the +3 state is highly oxidising and it reverts back to the more stable +1 state.
संबंधित प्रश्न
If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
Aluminium trifluoride is insoluble in anhydrous HF but dissolves on the addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous BF3 is bubbled through. Give reasons.
What happens when BF3 is reacted with ammonia?
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Write a balanced equation for B2H6 + NH3 → ?
A compound X, of boron reacts with NH3 on heating to give another compound Y which is called inorganic benzene. The compound X can be prepared by treating BF3 with Lithium aluminium hydride. The compounds X and Y are represented by the formulas.
Dry ice is ______.
Cement, the important building material is a mixture of oxides of several elements. Besides calcium, iron and sulphur, oxides of elements of which of the group (s) are present in the mixture?
Which of the following statements are correct. Answer on the basis of Figure.
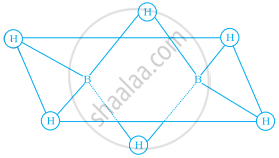
(i) The two birdged hydrogen atoms and the two boron atoms lie in one plane;
(ii) Out of six B – H bonds two bonds can be described in terms of 3 centre 2-electron bonds.
(iii) Out of six B – H bonds four B – H bonds can be described in terms of 3 centre 2 electron bonds;
(iv) The four-terminal B – H bonds are two centre-two electron regular bonds.
Explain the following:
Boron does not exist as B3+ ion.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Match the species given in Column I with properties given in Column II.
| Column I | Column II |
| (i) Diborane | (a) Used as a flux for soldering metals |
| (ii) Galluim | (b) Crystalline form of silica |
| (iii) Borax | (c) Banana bonds |
| (iv) Aluminosilicate | (d) Low melting, high boiling, useful for measuring high temperatures |
| (v) Quartz | (e) Used as catalyst in petrochemical industries |
Match the species given in Column I with the hybridisation given in Column II.
| Column I | Column II |
| (i) Boron in [B(OH)4]– | (a) sp2 |
| (ii) Aluminium in [Al(H2O)6]3+ | (b) sp3 |
| (iii) Boron in B2H6 | (c) sp3d2 |
| (iv) Carbon in Buckminsterfullerene | |
| (v) Silicon in \[\ce{SiO^{4-}4}\] | |
| (vi) Germanium in [GeCl6]2– |
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Atomic size
Account for the following observations:
Though fluorine is more electronegative than chlorine yet BF3 is a weaker Lewis acid than BCl3
Three pairs of compounds are given below. Identify that compound in each of the pairs which has group 13 element in more stable oxidation state. Give reason for your choice. State the nature of bonding also.
InCl3, InCl
A nonmetallic element of group 13, used in making bullet proof vests is extremely hard solid of black colour. It can exist in many allotropic forms and has unusually high melting point. Its trifluoride acts as Lewis acid towards ammonia. The element exihibits maximum covalency of four. Identify the element and write the reaction of its trifluoride with ammonia. Explain why does the trifluoride act as a Lewis acid.
Boron compounds behave as Lewis acids because of their ______.
Which one of the following is the correct statement?
