Advertisements
Advertisements
Question
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
Advertisements
Solution
Einstein’s photoelectric equation is given as:
eV0 = hv − `phi_0`
`"V"_0 = "h"/"e" "v" - phi_0/"e"` .............(1)
Where,
V0 = Stopping potential
h = Planck’s constant
e = Charge on an electron
v = Frequency of radiation
`phi_0` = Work function of a material
It can be concluded from equation (1) that potential V0 is directly proportional to frequency v.
Frequency is also given by the relation:
`"v" = "Speed of light (c)"/"Wavelenght (λ)"`
This relation can be used to obtain the frequencies of the various lines of the given wavelengths.
`"v"_1 = "c"/lambda_1 = (3 xx 10^8)/(3650 xx 10^(-10)) = 8.219 xx 10^14 "Hz"`
`"v"_2 = "c"/lambda_2 = (3 xx10^8)/(4047 xx 10^(-10)) = 7.412 xx 10^14 "Hz"`
`"v"_3 = "c"/lambda_3 = (3 xx 10^8)/(4358 xx 10^(-10)) = 6.884 xx 10^14 "Hz"`
`"v"_4 = "c"/lambda_4 = (3 xx 10^8)/(5461 xx 10^(-10)) = 5.493 xx 10^14 "Hz"`
`"v"_5 = "c"/lambda_5 = (3xx10^8)/(6907 xx 10^(-10)) = 4.343 xx 10^14 "Hz"`
The given quantities can be listed in tabular form as:
| Frequency × 1014 Hz | 8.219 | 7.412 | 6.884 | 5.493 | 4.343 |
| Stopping potential V0 | 1.28 | 0.95 | 0.74 | 0.16 | 0 |
The following figure shows a graph between νand V0.
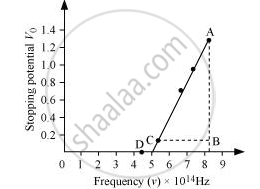
It can be observed that the obtained curve is a straight line. It intersects the ν-axis at 5 × 1014 Hz, which is the threshold frequency (v0) of the material. Point D corresponds to a frequency less than the threshold frequency. Hence, there is no photoelectric emission for the λ5 line, and therefore, no stopping voltage is required to stop the current.
Slope of the straight line = `"AB"/"CB" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
From equation (1), the slope `"h"/"e"` can be written as:
`"h"/"e" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
∴ `"h" = (1.12 xx 1.6 xx 10^(-19))/(2.726 xx 10^(14))`
= 6.573 × 10−34 Js
The work function of the metal is given as:
`phi_0` = hv0
= 6.573 × 10−34 × 5 × 1014
= 3.286 × 10−19 J
= `(3.286 xx 10^(-19))/(1.6 xx 1^(-18))`
= 2.054 eV
APPEARS IN
RELATED QUESTIONS
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Monochromatic radiation of wavelength 640.2 nm (1 nm = 10−9 m) from a neon lamp irradiates photosensitive material made of caesium on tungsten. The stopping voltage is measured to be 0.54 V. The source is replaced by an iron source and its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
The following graph shows the variation of photocurrent for a photosensitive metal :
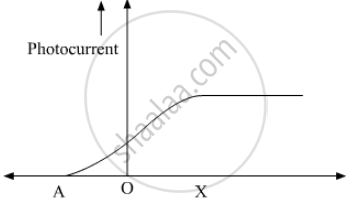
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
It is found that yellow light does not eject photoelectrons from a metal. Is it advisable to try with orange light or with green light?
Two photons of
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
If the wavelength of light in an experiment on photoelectric effect is doubled,
(a) photoelectric emission will not take place
(b) photoelectric emission may or may not take place
(c) the stopping potential will increase
(d) the stopping potential will decrease
Show that it is not possible for a photon to be completely absorbed by a free electron.
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
A small piece of cesium metal (φ = 1.9 eV) is kept at a distance of 20 cm from a large metal plate with a charge density of 1.0 × 10−9 C m−2 on the surface facing the cesium piece. A monochromatic light of wavelength 400 nm is incident on the cesium piece. Find the minimum and maximum kinetic energy of the photoelectrons reaching the large metal plate. Neglect any change in electric field due to the small piece of cesium present.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Define the term: threshold frequency
Explain how does (i) photoelectric current and (ii) kinetic energy of the photoelectrons emitted in a photocell vary if the frequency of incident radiation is doubled, but keeping the intensity same?
Show the graphical variation in the above two cases.
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
If photons of ultraviolet light of energy 12 eV are incident on a metal surface of work function of 4 eV, then the stopping potential (in eV) will be :
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
The figure shows a plot of stopping potential (V0) versus `1/lambda`, where λ is the wavelength of the radiation causing photoelectric emission from a surface. The slope of the line is equal to ______.

Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
