Advertisements
Advertisements
Question
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will ______.
Options
be doubled
be halved
become more than double
become less than double
Advertisements
Solution
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will become more than double.
Explanation:
According to Einstein's equation of the photoelectric effect,
`eV_0 = hv - varphi`
⇒ `V_0 = (hv - varphi)/e` ....(1)
Here, V0 = stopping potential
v = frequency of light
`varphi` = work function
Let the new frequency of light be 2ν and the corresponding stopping potential be V0'.
Therefore,
`eV_0^' = 2hv - varphi`
`V_0^' = (2hv - varphi)/e` ....(2)
Multiplying both sides of equation (1) by 2, we get:
`2V_0 = (2hv - 2varphi)/e` ....(3)
Now if we compare (2) and (3), it can be observed that: `(2hv - varphi)/e > (2hv - 2varphi)/e`
⇒ `V_0^' > 2V_0`
It is clear from the above equation that if the frequency of light in a photoelectric experiment is doubled, the stopping potential will be more than doubled.
APPEARS IN
RELATED QUESTIONS
Define the term 'intensity of radiation' in terms of photon picture of light.
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
The following graph shows the variation of photocurrent for a photosensitive metal :
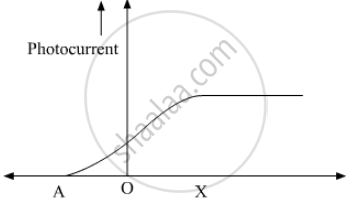
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Should the energy of a photon be called its kinetic energy or its internal energy?
The threshold wavelength of a metal is λ0. Light of wavelength slightly less than λ0 is incident on an insulated plate made of this metal. It is found that photoelectrons are emitted for some time and after that the emission stops. Explain.
If an electron has a wavelength, does it also have a colour?
The work function of a metal is hv0. Light of frequency v falls on this metal. Photoelectric effect will take place only if
A photon of energy hv is absorbed by a free electron of a metal with work-function hv − φ.
Show that it is not possible for a photon to be completely absorbed by a free electron.
Find the maximum magnitude of the linear momentum of a photoelectron emitted when a wavelength of 400 nm falls on a metal with work function 2.5 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a monochromatic beam is 1.2 × 1015 times per second. Find the maximum kinetic energy of the photoelectrons when this light falls on a metal surface whose work function is 2.0 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Define the term: threshold frequency
Do all the electrons that absorb a photon come out as photoelectrons?
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Consider a 20 W bulb emitting light of wavelength 5000 Å and shining on a metal surface kept at a distance 2 m. Assume that the metal surface has work function of 2 eV and that each atom on the metal surface can be treated as a circular disk of radius 1.5 Å.
- Estimate no. of photons emitted by the bulb per second. [Assume no other losses]
- Will there be photoelectric emission?
- How much time would be required by the atomic disk to receive energy equal to work function (2 eV)?
- How many photons would atomic disk receive within time duration calculated in (iii) above?
- Can you explain how photoelectric effect was observed instantaneously?
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
If photons of ultraviolet light of energy 12 eV are incident on a metal surface of work function of 4 eV, then the stopping potential (in eV) will be :
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
