Advertisements
Advertisements
Question
Consider a 20 W bulb emitting light of wavelength 5000 Å and shining on a metal surface kept at a distance 2 m. Assume that the metal surface has work function of 2 eV and that each atom on the metal surface can be treated as a circular disk of radius 1.5 Å.
- Estimate no. of photons emitted by the bulb per second. [Assume no other losses]
- Will there be photoelectric emission?
- How much time would be required by the atomic disk to receive energy equal to work function (2 eV)?
- How many photons would atomic disk receive within time duration calculated in (iii) above?
- Can you explain how photoelectric effect was observed instantaneously?
Advertisements
Solution
According to the problem, P = 20 W, λ = 5000 Å = 5000 × 10–10 m, distance (d) = 2 m, work function `phi_0` = 2 eV, radius r = 1.5 Å = 1.5 × 10–10 m
Now, Number of photon emitted by bulb per second, n' = `(dN)/(dt)`
i. Number of photon emitted by bulb per second is n’ = `(P)/((hc)/λ) = (Pλ)/(hc)`
= `(20 xx (5000 xx 10^-10))/((6.62 xx 10^-34) xx (3 xx 10^8))`
⇒ n' = 5 × 1019/sec
ii. Energy of the incident photon = `(hc)/λ`
= `((6.62 xx 10^-34) xx (3 xx 10^8))/(5000 xx 10^-10 xx 1.6 xx 10^-19)`
= 2.48 ev
As this energy is greater than 2 eV (i.e., a work function of the metal surface), hence photoelectric emission takes place.
iii. Let Δt be the time spent in getting the energy `phi` = (work function of metal).
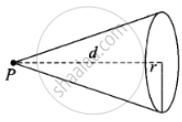
Consider the figure, if P is the power of source then energy received by the atomic disc
`p/(4πd^2) xx pir^2Δt = phi_0`
⇒ Δt = `(4phi_0d^2)/(Pr^2)`
= `(4 xx (2 xx 1.6 xx 10^-19) xx 2^2)/(20 xx (1.5 xx 10^-10)^2`
= 2.84 s
iv. Number of photons received by the atomic disc in time Δt is
N = `(n^' xx pir^2)/(4pid^2) xx Δt`
= `(n^'r^2Δt)/(4d^2)`
= `((5 xx 10^19) xx (1.5 xx 10^-10)^2 xx 28.4)/(4 xx (2)^2`
= 2
Now let us discuss the last part in detail. As the time of emission of electrons is 11.04 s.
v. In photoelectric emission, there is a collision between the incident photon and free electron of the metal surface, which lasts for a very short interval of time (≈ 10–9 s), hence we say photoelectric emission is instantaneous.
APPEARS IN
RELATED QUESTIONS
Monochromatic radiation of wavelength 640.2 nm (1 nm = 10−9 m) from a neon lamp irradiates photosensitive material made of caesium on tungsten. The stopping voltage is measured to be 0.54 V. The source is replaced by an iron source and its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
Can we find the mass of a photon by the definition p = mv?
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
Should the energy of a photon be called its kinetic energy or its internal energy?
If an electron has a wavelength, does it also have a colour?
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
When stopping potential is applied in an experiment on photoelectric effect, no photoelectric is observed. This means that
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
When the sun is directly overhead, the surface of the earth receives 1.4 × 103 W m−2 of sunlight. Assume that the light is monochromatic with average wavelength 500 nm and that no light is absorbed in between the sun and the earth's surface. The distance between the sun and the earth is 1.5 × 1011 m. (a) Calculate the number of photons falling per second on each square metre of earth's surface directly below the sun. (b) How many photons are there in each cubic metre near the earth's surface at any instant? (c) How many photons does the sun emit per second?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The work function of a metal is 2.5 × 10−19 J. (a) Find the threshold frequency for photoelectric emission. (b) If the metal is exposed to a light beam of frequency 6.0 × 1014 Hz, what will be the stopping potential?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
In the case of photoelectric effect experiment, explain the following facts, giving reasons.
The photoelectric current increases with increase of intensity of incident light.
In photoelectric effect the photo current ______.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
What is the effect of threshold frequency and stopping potential on increasing the frequency of the incident beam of light? Justify your answer.
The difference between threshold wavelengths for two metal surfaces A and B having work function ΦA = 9 eV and ΦB = 4.5 eV in nm is ______.
(Given, hc = 1242 eV nm)
