Advertisements
Advertisements
प्रश्न
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
Advertisements
उत्तर
Einstein’s photoelectric equation is given as:
eV0 = hv − `phi_0`
`"V"_0 = "h"/"e" "v" - phi_0/"e"` .............(1)
Where,
V0 = Stopping potential
h = Planck’s constant
e = Charge on an electron
v = Frequency of radiation
`phi_0` = Work function of a material
It can be concluded from equation (1) that potential V0 is directly proportional to frequency v.
Frequency is also given by the relation:
`"v" = "Speed of light (c)"/"Wavelenght (λ)"`
This relation can be used to obtain the frequencies of the various lines of the given wavelengths.
`"v"_1 = "c"/lambda_1 = (3 xx 10^8)/(3650 xx 10^(-10)) = 8.219 xx 10^14 "Hz"`
`"v"_2 = "c"/lambda_2 = (3 xx10^8)/(4047 xx 10^(-10)) = 7.412 xx 10^14 "Hz"`
`"v"_3 = "c"/lambda_3 = (3 xx 10^8)/(4358 xx 10^(-10)) = 6.884 xx 10^14 "Hz"`
`"v"_4 = "c"/lambda_4 = (3 xx 10^8)/(5461 xx 10^(-10)) = 5.493 xx 10^14 "Hz"`
`"v"_5 = "c"/lambda_5 = (3xx10^8)/(6907 xx 10^(-10)) = 4.343 xx 10^14 "Hz"`
The given quantities can be listed in tabular form as:
| Frequency × 1014 Hz | 8.219 | 7.412 | 6.884 | 5.493 | 4.343 |
| Stopping potential V0 | 1.28 | 0.95 | 0.74 | 0.16 | 0 |
The following figure shows a graph between νand V0.
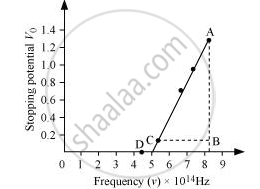
It can be observed that the obtained curve is a straight line. It intersects the ν-axis at 5 × 1014 Hz, which is the threshold frequency (v0) of the material. Point D corresponds to a frequency less than the threshold frequency. Hence, there is no photoelectric emission for the λ5 line, and therefore, no stopping voltage is required to stop the current.
Slope of the straight line = `"AB"/"CB" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
From equation (1), the slope `"h"/"e"` can be written as:
`"h"/"e" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
∴ `"h" = (1.12 xx 1.6 xx 10^(-19))/(2.726 xx 10^(14))`
= 6.573 × 10−34 Js
The work function of the metal is given as:
`phi_0` = hv0
= 6.573 × 10−34 × 5 × 1014
= 3.286 × 10−19 J
= `(3.286 xx 10^(-19))/(1.6 xx 1^(-18))`
= 2.054 eV
APPEARS IN
संबंधित प्रश्न
(a) Estimate the speed with which electrons emitted from a heated emitter of an evacuated tube impinge on the collector maintained at a potential difference of 500 V with respect to the emitter. Ignore the small initial speeds of the electrons. The specific charge of the electron, i.e., its e/m is given to be 1.76 × 1011 C kg−1.
(b) Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
The following graph shows the variation of photocurrent for a photosensitive metal :
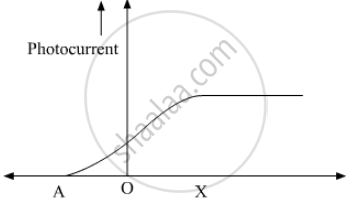
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
Two photons of
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will ______.
A photon of energy hv is absorbed by a free electron of a metal with work-function hv − φ.
If the wavelength of light in an experiment on photoelectric effect is doubled,
(a) photoelectric emission will not take place
(b) photoelectric emission may or may not take place
(c) the stopping potential will increase
(d) the stopping potential will decrease
In which of the following situations, the heavier of the two particles has smaller de Broglie wavelength? The two particles
(a) move with the same speed
(b) move with the same linear momentum
(c) move with the same kinetic energy
(d) have fallen through the same height
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Calculate the number of photons emitted per second by a 10 W sodium vapour lamp. Assume that 60% of the consumed energy is converted into light. Wavelength of sodium light = 590 nm
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A totally reflecting, small plane mirror placed horizontally faces a parallel beam of light, as shown in the figure. The mass of the mirror is 20 g. Assume that there is no absorption in the lens and that 30% of the light emitted by the source goes through the lens. Find the power of the source needed to support the weight of the mirror.

(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Show that it is not possible for a photon to be completely absorbed by a free electron.
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A small piece of cesium metal (φ = 1.9 eV) is kept at a distance of 20 cm from a large metal plate with a charge density of 1.0 × 10−9 C m−2 on the surface facing the cesium piece. A monochromatic light of wavelength 400 nm is incident on the cesium piece. Find the minimum and maximum kinetic energy of the photoelectrons reaching the large metal plate. Neglect any change in electric field due to the small piece of cesium present.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Consider a thin target (10–2 cm square, 10–3 m thickness) of sodium, which produces a photocurrent of 100 µA when a light of intensity 100W/m2 (λ = 660 nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
The work function for a metal surface is 4.14 eV. The threshold wavelength for this metal surface is ______.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
Which of the following options represents the variation of photoelectric current with property of light shown on the x-axis?