Advertisements
Advertisements
प्रश्न
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
Advertisements
उत्तर
Einstein’s photoelectric equation is given as:
eV0 = hv − `phi_0`
`"V"_0 = "h"/"e" "v" - phi_0/"e"` .............(1)
Where,
V0 = Stopping potential
h = Planck’s constant
e = Charge on an electron
v = Frequency of radiation
`phi_0` = Work function of a material
It can be concluded from equation (1) that potential V0 is directly proportional to frequency v.
Frequency is also given by the relation:
`"v" = "Speed of light (c)"/"Wavelenght (λ)"`
This relation can be used to obtain the frequencies of the various lines of the given wavelengths.
`"v"_1 = "c"/lambda_1 = (3 xx 10^8)/(3650 xx 10^(-10)) = 8.219 xx 10^14 "Hz"`
`"v"_2 = "c"/lambda_2 = (3 xx10^8)/(4047 xx 10^(-10)) = 7.412 xx 10^14 "Hz"`
`"v"_3 = "c"/lambda_3 = (3 xx 10^8)/(4358 xx 10^(-10)) = 6.884 xx 10^14 "Hz"`
`"v"_4 = "c"/lambda_4 = (3 xx 10^8)/(5461 xx 10^(-10)) = 5.493 xx 10^14 "Hz"`
`"v"_5 = "c"/lambda_5 = (3xx10^8)/(6907 xx 10^(-10)) = 4.343 xx 10^14 "Hz"`
The given quantities can be listed in tabular form as:
| Frequency × 1014 Hz | 8.219 | 7.412 | 6.884 | 5.493 | 4.343 |
| Stopping potential V0 | 1.28 | 0.95 | 0.74 | 0.16 | 0 |
The following figure shows a graph between νand V0.
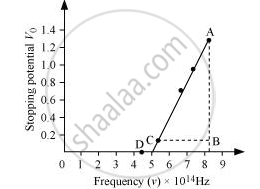
It can be observed that the obtained curve is a straight line. It intersects the ν-axis at 5 × 1014 Hz, which is the threshold frequency (v0) of the material. Point D corresponds to a frequency less than the threshold frequency. Hence, there is no photoelectric emission for the λ5 line, and therefore, no stopping voltage is required to stop the current.
Slope of the straight line = `"AB"/"CB" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
From equation (1), the slope `"h"/"e"` can be written as:
`"h"/"e" = (1.28 - 0.16)/((8.214 - 5.493) xx 10^14)`
∴ `"h" = (1.12 xx 1.6 xx 10^(-19))/(2.726 xx 10^(14))`
= 6.573 × 10−34 Js
The work function of the metal is given as:
`phi_0` = hv0
= 6.573 × 10−34 × 5 × 1014
= 3.286 × 10−19 J
= `(3.286 xx 10^(-19))/(1.6 xx 1^(-18))`
= 2.054 eV
APPEARS IN
संबंधित प्रश्न
Light of intensity 10−5 W m−2 falls on a sodium photo-cell of surface area 2 cm2. Assuming that the top 5 layers of sodium absorb the incident energy, estimate time required for photoelectric emission in the wave-picture of radiation. The work function for the metal is given to be about 2 eV. What is the implication of your answer?
Can we find the mass of a photon by the definition p = mv?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
Can a photon be deflected by an electric field? Or by a magnetic field?
Planck's constant has the same dimensions as
Two photons of
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
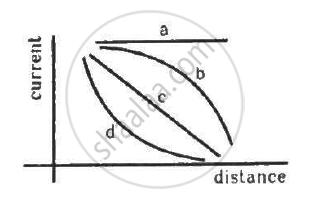
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
Photoelectric effect supports quantum nature of light because
(a) there is a minimum frequency below which no photoelectrons are emitted
(b) the maximum kinetic energy of photoelectrons depends only on the frequency of light and not on its intensity
(c) even when the metal surface is faintly illuminated the photoelectrons leave the surface immediately
(d) electric charge of the photoelectrons is quantised
An atom absorbs a photon of wavelength 500 nm and emits another photon of wavelength 700 nm. Find the net energy absorbed by the atom in the process.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
When the sun is directly overhead, the surface of the earth receives 1.4 × 103 W m−2 of sunlight. Assume that the light is monochromatic with average wavelength 500 nm and that no light is absorbed in between the sun and the earth's surface. The distance between the sun and the earth is 1.5 × 1011 m. (a) Calculate the number of photons falling per second on each square metre of earth's surface directly below the sun. (b) How many photons are there in each cubic metre near the earth's surface at any instant? (c) How many photons does the sun emit per second?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In an experiment on photoelectric effect, the stopping potential is measured for monochromatic light beams corresponding to different wavelengths. The data collected are as follows:-
Wavelength (nm): 350 400 450 500 550
Stopping potential (V): 1.45 1.00 0.66 0.38 0.16
Plot the stopping potential against inverse of wavelength (1/λ) on a graph paper and find (a) Planck's constant (b) the work function of the emitter and (c) the threshold wavelength.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The figure is the plot of stopping potential versus the frequency of the light used in an experiment on photoelectric effect. Find (a) the ratio h/e and (b) the work function.

Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
- Assertion (A): For the radiation of a frequency greater than the threshold frequency, the photoelectric current is proportional to the intensity of the radiation.
- Reason (R): Greater the number of energy quanta available, the greater the number of electrons absorbing the energy quanta and the greater the number of electrons coming out of the metal.
