Advertisements
Advertisements
प्रश्न
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Advertisements
उत्तर
Effect of intensity: If the intensity of light is increased (while its frequency is kept the same) the current levels off at a higher value, showing that more electrons are being emitted per unit of time. But the stopping potential V0 doesn't change, i.e. Intensity `∝` no . of incident photon no. of emitted photoelectron per time photocurrent.
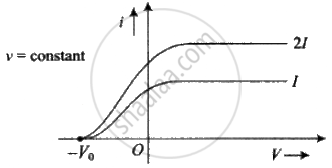
Effect of frequency: If the frequency of incident light increases, (keeping intensity constant) stopping potential increases but there is no change in photoelectric current.
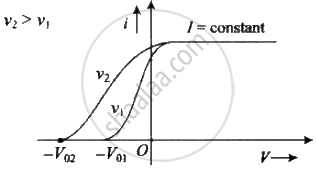
Let us assume nA is the number of photons falling per second of beam A and nB is the number of photons falling per second of beam B.
And it is given that the number of photons hitting the screen by beam A is twice that by beam B.nA = 2nB
The energy of the falling photon of beam A = hvA
The energy of a falling photon of beam B = hvB
Now, according to the question, the intensity of A is equal to the intensity of B.
Therefore, I = nAvA = nBvB
⇒ `v_A/v_B = n_B/n_A = n_B/(2n_B) = 1/2`
⇒ vB = 2vA
APPEARS IN
संबंधित प्रश्न
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Every metal has a definite work function. Why do all photoelectrons not come out with the same energy if incident radiation is monochromatic? Why is there an energy distribution of photoelectrons?
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
Should the energy of a photon be called its kinetic energy or its internal energy?
It is found that photosynthesis starts in certain plants when exposed to sunlight, but it does not start if the plants are exposed only to infrared light. Explain.
Planck's constant has the same dimensions as
The work function of a metal is hv0. Light of frequency v falls on this metal. Photoelectric effect will take place only if
Photoelectric effect supports quantum nature of light because
(a) there is a minimum frequency below which no photoelectrons are emitted
(b) the maximum kinetic energy of photoelectrons depends only on the frequency of light and not on its intensity
(c) even when the metal surface is faintly illuminated the photoelectrons leave the surface immediately
(d) electric charge of the photoelectrons is quantised
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A beam of white light is incident normally on a plane surface absorbing 70% of the light and reflecting the rest. If the incident beam carries 10 W of power, find the force exerted by it on the surface.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, Show that the force on the sphere due to the light falling on it is the same even if the sphere is not perfectly absorbing.
Show that it is not possible for a photon to be completely absorbed by a free electron.
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
Explain how does (i) photoelectric current and (ii) kinetic energy of the photoelectrons emitted in a photocell vary if the frequency of incident radiation is doubled, but keeping the intensity same?
Show the graphical variation in the above two cases.
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
The work function for a metal surface is 4.14 eV. The threshold wavelength for this metal surface is ______.
Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
