Topics
Electric Charges and Fields
- Electric Charge
- Positive and Negative Charges
- Electron Theory of Electrification
- Conductors and Insulators
- Electrostatic Induction
- Important Properties of Electric Charge
- Scalar Form of Coulomb’s Law
- Coulomb's Law in Vector Form
- Principle of Superposition
- Equilibrium of Charge and System of Charges
- Electric Field
- Intensity of Electric Field
- Electric Field Intensity Due to a Point-Charge
- Intensity of Electric Field due to a Continuous Charge Distribution
- Electric Lines of Force
- Electric Dipole
- Electric Field due to an Electric Dipole
- Torque on a Dipole in a Uniform Electric Field
- Motion of a Charged Particle in Uniform Electric Field
Electrostatics
Current Electricity
Gauss' Theorem
- Area Vector
- Solid Angle
- Electric Flux
- Gauss' Theorem
- Gaussian Surface and its Properties
- Applications of Gauss' Theorem > Electric Field due to a Point Charge
- Applications of Gauss' Theorem > Electric Field due to an Infinite Line of Charge
- Applications of Gauss' Theorem > Electric Field due to an Infinite Plane Sheet of Charge
- Applications of Gauss' Theorem > Electric Field due to Two Infinite Parallel Sheets of Charge
- Applications of Gauss' Theorem > Electric Field Intensity Just Outside a Charged Conductor
- Applications of Gauss' Theorem > Electric Field due to a Uniformly Charged Thin Spherical Shell
- Applications of Gauss' Theorem > Electric Field due to a Uniformly Charged Sphere
- Overview: Gauss' Theorem
Electric Potential
- Electric Potential
- Potential and Potential Difference
- Electron-volt or eV
- Electric Potential Due to a Point Charge
- Potential due to a Group of Point Charges
- Potential Gradient
- Electric Field as Gradient of Electric Potential: Relation between E and V
- Electric Potential Energy of a System of Charges
- Equipotential Surfaces
- Potential Due to an Electric Dipole
- Work Done in Rotating an Electric Dipole in an Electric Field
- Electric Potential Energy of an Electric Dipole in an Electrostatic Field
- Overview: Electric Potential
Magnetic Effects of Current and Magnetism
Capacitors and Dielectrics
- Conductors and Insulators
- Free Charges and Bound Charges Inside a Conductor
- Capacitance of a Conductor
- Capacitance of an Isolated Spherical Conductor
- Potential Energy of a Charged Conductor
- Redistribution of Charges: Common Potential
- Capacitors
- Capacitance of a Capacitor
- Expression for Capacitance of a Parallel-Plate Capacitor
- Dependence of the Capacitance of a Capacitor
- Capacitance of a Parallel-Plate Capacitor with Dielectric Slab between Plates
- Combination of Capacitors
- Energy Stored in a Charged Capacitor
- Force between the Plates of a Charged Parallel-Plate Capacitor
- Induced Charges in a Dielectric Slab in a Capacitor
- Dielectrics
- Electric Polarisation of Matter
- Effect of Introducing a Dielectric between the Plates of a Charged Capacitor
- Overview: Capacitors and Dielectrics
Electromagnetic Induction and Alternating Currents
Electric Resistance and Ohm's Law
- Electric Current
- Current Density
- Mechanism of Flow of Charge in Metals
- Transport Properties of Free Electrons
- Mobility of Electrons
- Electric Current and Its Related Concepts
- Resistance
- Ohm's Law
- Experimental Verification of Ohm’s Law and Ohmic Resistors
- Exceptions of Ohm's Law : Non-Linear V-I Characteristics
- Dynamic Resistance
- Derivation of Ohm's Law
- Specific Resistance or Electrical Resistivity
- Ohm's law in Vector Form
- Dependence of Resistance on the Dimensions of the Conductor
- Effect of Temperature on Resistivity
- Colour Code of Carbon Resistors
- Combinations of Resistances
- On the Basis of Series and Parallel Combinations, to Prove R ∝ 1/A
- Electric Energy and Power
- Commercial Units of Electricity Consumption
- Net Power Consumption
- Overview: Electric Resistance and Ohm's Law
Electromagnetic Waves
DC Circuits and Measurements
- Electric cell
- Electromotive Force (emf)
- Terminal Potential Difference
- Internal Resistance of a Cell
- Relation between E, V, and r
- Combinations of Cells
- Kirchhoff’s Laws
- Wheatstone Bridge
- Rheostat
- Metre Bridge: Slide-Wire Bridge
- Potentiometer
- Overview: DC Circuits and Measurements
Optics (Ray and Wave Optics)
Dual Nature of Radiation and Matter
Moving Charges and Magnetic Field
- Magnetic Field
- Oersted's Experiment
- Biot-Savart Law
- Comparison of Coulomb's Law and Biot-Savart's Law
- Rules to Determine the Direction of Developed Magnetic Field
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field on the Axis of a Circular Current-Carrying Loop
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Ampere’s Circuital Law
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Thin Wire
- Applications of Ampere’s Circuital Law > Magnetic Field of a Toroidal Solenoid
- Force on a Moving Charge in a Uniform Magnetic Field
- Magnetic Field Defined by Magnetic Force
- Motion of Charged Particles in a Uniform Magnetic Field
- Lorentz Force
- Cyclotron
- Force on a Current Carrying Conductor in a Magnetic Field
- Ampere: Based on Force Between Currents
- Overview: Moving Charges and Magnetic Field
Atoms and Nuclei
Torque on a Current-Loop : Moving-Coil Galvanometer
Magnetic Field and Earth's Magnetism
- Current Loop as a Magnetic Dipole
- Magnetic Dipole Moment of a Revolving Electron
- Magnetic Field of a Magnetic Dipole (Small Bar Magnet)
- Torque on a Magnetic Dipole (Bar Magnet) in a Uniform Magnetic Field
- Potential Energy of a Magnet in a Magnetic Field
- Current-Carrying Solenoid as an Equivalent to a Bar Magnet
- Magnetic Lines of Force
- Earth’s Magnetic Field
- Elements of the Earth's Magnetic Field > Angle of Declination
- Elements of the Earth's Magnetic Field > Angle of Dip or Magnetic Inclination
- Elements of the Earth's Magnetic Field > Horizontal Component of Earth's Magnetic Field
- Overview: Magnetic Field and Earth's Magnetism
Electronic Devices
Communication Systems
Magnetic Classification of Substances
- Classification of Substances According to their Magnetic Behaviour
- Terms Used in Magnetism
- Properties of Dia-, Para-, and Ferromagnetic Substances
- Explanation of Dia-, Para-, and Ferromagnetism based on the Atomic Model of Magnetism
- Hysteresis: Retentivity and Coercivity
- Differences in Magnetic Properties of Soft Iron and Steel
- Magnetic Materials
- Overview: Magnetic Classification of Substances
Electromagnetic Induction
- Magnetic Flux
- Electromagnetic Induction
- Faraday's Laws of Electromagnetic Induction
- Induced Current and Induced Charge
- Methods of Changing the Magnetic Flux
- Motion of a Straight Conductor in a Uniform Magnetic Field (Motional EMF)
- Explanation of Electromagnetic Induction in Terms of Lorentz Force: Proof of Faraday's Law
- Motional emf in Rotating a Conducting Rod in a Uniform Magnetic Field
- Self – Induction
- Self-Inductance of a Long Solenoid
- Energy Stored in an Inductor
- Examples of the Effects of Self-Induced Current
- Mutual Induction
- Mutual Inductance
- Eddy Currents or Foucault Currents
- Overview: Electromagnetic Induction
Alternating Current
- Alternating Voltage and Current in a Rotating Coil
- Definitions Regarding Alternating Voltage and Current
- Mean (or Average) Value of Alternating Current (or Voltage)
- Root-Mean-Square Value of Alternating Current
- Phasors and Phasor Diagrams
- Types of AC Circuits
- Circuit containing Resistance Only
- Circuit containing Inductance Only
- Circuit containing Capacitance Only
- Circuit containing Inductance and Resistance in Series (L-R Series Circuit)
- Circuit containing Capacitance and Resistance in Series (C-R Series Circuit)
- Circuit containing Inductance and Capacitance (L-C Circuit)
- Circuit containing Inductance, Capacitance and Resistance in Series (L-C-R Series Circuit)
- Power in AC Circuit
- Wattless Current
- Half Power Points, Bandwidth and Q-Factor
- Choke Coil
- Electrical Oscillations in L-C Circuit
- Resonant Circuits
- Frequency Response of AC Circuits
- A.C. Generator
- Transformers
- Utility of Alternating Current in Comparison to Direct Current
- Overview: Alternating Current
Electromagnetic Waves
- Displacement Current
- Relation between Conduction and Displacement Current
- Maxwell's Equation
- Concept of Electromagnetic Waves
- Field Magnitude Relation in Free Space
- Energy Density in Electromagnetic Waves
- Transverse Nature of Electromagnetic Waves
- Electromagnetic Spectrum
- Overview: Electromagnetic Waves
Reflection of Light: Spherical Mirrors
- Spherical Mirrors
- Fundamental Terms Related to Spherical Mirrors
- Relation Between Focal Length and Radius of Curvature of a Spherical Mirror
- Rules to Trace the Image Formed by Spherical Mirrors
- Conditions of Image Formation
- Position and Nature of Image Formed by Spherical Mirrors
- Sign Convention
- Mirror Formula for Concave Mirror
- Mirror Formula for Convex Mirror
- Linear Magnification by Spherical Mirrors
- Uses of Spherical Mirrors
- Overview: Reflection of Light: Spherical Mirrors
Refraction of Light at a Plane Interface : Total Internal Reflection : Optical Fibre
- Refraction of Light
- Laws of Refraction
- Cause of Refraction
- Physical Significance of Refractive Index
- Reversibility of Light
- Refraction of Light Through a Rectangular Glass Block
- Refraction through Parallel Multiple Media
- Real and Apparent Depths: Normal Displacement
- Critical Angle
- Total Internal Reflection
- Applications of Total Internal Reflection
- Overview: Refraction of Light at a Plane Interface
Refraction of Light at Spherical Surfaces : Lenses
- Coordinate Geometry Sign Convention for Measuring Distances and Lengths
- Refraction at Concave Spherical Surface
- Refraction at a Convex Spherical Surface
- Concept of Lenses
- Converging and Diverging Actions of Lenses
- Lens Maker's Formula
- Factors Affecting Focal Length of a Lens
- Image Formation by Thin Lenses
- Ray Diagrams for Formation of Image by a Convex Lens
- Ray Diagram for Formation of Image by a Concave Lens
- Linear Magnification by Spherical Lenses
- Power of a Lens
- Combined Focal Length of Two Thin Lenses in Contact
- Combination of Lenses and Mirrors
- Overview: Refraction of Light at Spherical Surfaces: Lenses
Refraction and Dispersion of Light through a Prism
Optical Instruments
Wave Nature of Light : Huygens' Principle
Interference of Light
Diffraction of Light
Polarisation of Light
Photoelectric Effect
Matter Waves
X-Rays
Atom, Origin of Spectra : Bohr's Theory of Hydrogen Atom
Nuclear Structure
Radioactivity
Mass-Energy Equivalence : Nuclear Binding Energy
Nuclear Fission and Nuclear Fusion : Sources of Energy
Semiconductor Electronics
Junction Diodes
Junction Transistors
Logic Gates
Communication Systems
Definition: Photoelectric Effect
The phenomenon of emission of electronsfrom metals under the effect of light is called 'photoelectric effect'.
Definition: Stopping Potential
The negative potential of P2 (relative to P1) at which the photoelectric current becomes zero is called 'stopping potential' or 'cut-off potential'.
Definition: Threshold Frequency or Cut-off Frequency
The lowest frequency of light which can emit photoelectrons from a material is called the 'threshold frequency' or 'cut-off frequency' of that material.
Definition: Work Function
The minimum energy required for the emission of photoelectron from a metal is called the 'work function' of that metal.
Definition: Intensity of Wave
The energy crossing per unit area per unit time perpendicular to the direction of propagation of wave is called the intensity of wave.
Formula: Kinetic Mass of Photon
m = \[\frac {E}{c^2}\] = \[\frac {hv}{c^2}\] = \[\frac {h}{cλ}\]
Formula: Momentum of Photon
p = \[\frac {E}{c}\] = \[\frac {h}{λ}\]
Formula: Photon Emission Rate
n = \[\frac {Pλ}{h c}\]
Formula: Radiation Power (Photon Form)
P = nh\[\frac {c}{λ}\]
Law: Laws of Photoelectric Emission
- The rate of emission of photoelectrons from the surface of a metal varies directly as the intensity of the incident light falling on the surface.
- The maximum kinetic energy of the emitted photoelectrons is independent of the intensity of the incident light.
- The maximum kinetic energy of the photoelectrons increases linearly with an increase in the frequency of the incident light.
- If the frequency of the incident light is below a certain lowest value, then no photoelectron is emitted from the metal. This lowest frequency (threshold frequency) is different for different metals.
- As soon as the light is incident on the surface of the metal, the photoelectrons are emitted instantly; that is, there is no time-lag between the incidence of light and the emission of electrons.
Key Points: Photoelectric Effect
- Hertz (1887) observed that ultraviolet light makes electric discharge easier from a metal surface.
- Hallwachs’ experiment showed that current flows only when ultraviolet light strikes the negative plate, not the positive plate.
- J.J. Thomson (1898) proved that light falling on a metal surface causes the emission of electrons.
- Lenard (1900) explained that electrons emitted from the negative plate are attracted to the positive plate, producing current.
- Short-wavelength (high-frequency) light is more effective in producing photoelectric emission than long-wavelength light.
Key Points: Hertz and Lenard's Observations
- Photoelectric current increases with incident light intensity when the frequency is kept constant.
- For sufficiently high anode potential, the photoelectric current reaches a maximum (saturation current).
- Stopping potential is independent of light intensity and depends on the maximum kinetic energy of photoelectrons.
- A higher frequency of incident light produces photoelectrons with greater maximum kinetic energy.
- No photoelectric emission occurs below a certain frequency, regardless of the intensity or duration of light.
Key Points: Failure of Wave Theory
- Wave theory fails because it predicts that electron energy should increase with light intensity, but experiments show that it does not.
- Wave theory cannot explain the existence of a threshold frequency, below which no photoelectrons are emitted.
- Wave theory predicts a time lag in emission, but photoelectrons are emitted instantaneously.
Key Points: Planck's Photon Hypothesis
- Wave theory failed to explain experimental observations of the photoelectric effect.
- Black-body radiation contains all wavelengths, and classical theories could not explain its energy distribution.
- Planck proposed that radiation is emitted discontinuously in small energy packets, now known as quanta (photons).
- The energy of a photon is hνh\nuhν, and radiation energy is emitted only in integral multiples of hνh\nuhν.
- Einstein (1905) explained the photoelectric effect using Planck’s photon hypothesis.
Key Points: Photoelectric Equation
- Light consists of photons, each having energy hνh\nuhν; light intensity depends on the number of photons.
- A photon transfers its entire energy to a single electron during photoelectric emission.
- Part of the photon energy is used to overcome the work function, and the rest appears as kinetic energy of the electron.
- Electrons emitted from the metal surface have maximum kinetic energy because they experience no energy loss in collisions.
- Einstein’s photoelectric equation is
Ek = hν − W = h(ν − ν0) - Increasing light intensity increases the number of photoelectrons, but not their maximum kinetic energy.
- Photoelectric emission is instantaneous, and Einstein’s explanation fully accounts for all laws of the photoelectric effect.
Key Points: Determination of Planck's Constant
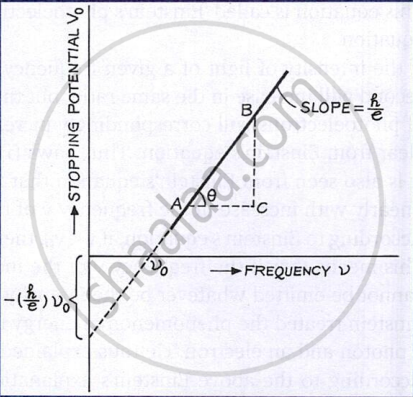
- A graph of stopping potential V0 versus frequency ν is a straight line, showing a linear relation between them.
- The slope of the V0 – ν graph equals h/e, hence Planck’s constant h can be determined using the known value of electronic charge e.
Key Points: Properties of Photons
- Radiation behaves like a stream of particles called photons during interaction with matter.
- Photons travel in straight lines at the speed of light.
- Each photon has energy E = hν = \[\frac {hc}{λ}\] and momentum p = \[\frac {E}{c}\].
- On a change of medium, the speed and wavelength of a photon change, but its frequency remains constant.
- Photon energy is independent of light intensity; higher intensity means more photons per second.
- A photon has zero rest mass, but an equivalent mass given by
- m = \[\frac {h}{cλ}\]
- Photons are electrically neutral and are not deflected by electric or magnetic fields.
- In photon–particle collisions, total energy and momentum are conserved.
- A photon retains its identity until absorbed by an atom, after which its identity is lost.
