Advertisements
Advertisements
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
Concept: undefined >> undefined
Can X-rays be used for photoelectric effect?
Concept: undefined >> undefined
Advertisements
Can X-rays be polarised?
Concept: undefined >> undefined
X-ray and visible light travel at the same speed in vacuum. Do they travel at the same speed in glass?
Concept: undefined >> undefined
Characteristic X-rays may be used to identify the element from which they are being emitted. Can continuous X-rays be used for this purpose?
Concept: undefined >> undefined
Is it possible that in a Coolidge tube characteristic Lα X-rays are emitted but not Kα X-rays?
Concept: undefined >> undefined
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
Concept: undefined >> undefined
Can a hydrogen atom emit characteristic X-rays?
Concept: undefined >> undefined
Why is exposure to X-rays injurious to health but not exposure to visible light, when both are electromagnetic waves?
Concept: undefined >> undefined
An X-ray beam can be deflected
Concept: undefined >> undefined
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
Concept: undefined >> undefined
The energy of a photon of a characteristic X-ray from a Coolidge tube comes from
Concept: undefined >> undefined
If the potential difference applied to the tube is doubled and the separation between the filament and the target is also doubled, the cutoff wavelength
Concept: undefined >> undefined
If the current in the circuit for heating the filament is increased, the cutoff wavelength
Concept: undefined >> undefined
Moseley's Law for characteristic X-ray is √v = a(Z − b). Here,
Concept: undefined >> undefined
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
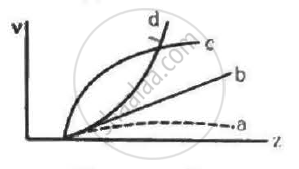
Concept: undefined >> undefined
The X-ray beam emerging from an X-ray tube
Concept: undefined >> undefined
One of the following wavelengths is absent and the rest are present in the X-rays coming from a Coolidge tube. Which one is the absent wavelength?
Concept: undefined >> undefined
The figure shows the intensity-wavelength relations of X-rays coming from two different Coolidge tubes. The solid curve represents the relation for the tube A in which the potential difference between the target and the filament is VA and the atomic number of the target material is ZA. These quantities are VB and ZB for the other tube. Then,

Concept: undefined >> undefined
50% of the X-rays coming from a Coolidge tube are able to pass through a 0.1 mm thick aluminium foil. If the potential difference between the target and the filament is increased, the fraction of the X-rays passing through the same foil will be
Concept: undefined >> undefined
