Definitions [29]
Define the following:
Chirality
The relationship between a chiral molecule and its mirror image is similar to the relationship between left and right hands. Therefore it is called handedness or chirality
Define the following:
Plane polarized light
A light having oscillations only in one plane perpendicular to direction of propagation of light is known as plane polarized light.
Define organometallic compound.
Active metals like sodium, magnesium cadmium readily combine with alkyl chlorides, bromides and iodides to form compounds containing carbon-metal bonds. These are known as organometallic compounds.
Halogen derivatives in which halogen is bonded to an sp³ hybridised carbon of an alkyl group are called haloalkanes or alkyl halides.
Isomers having the same bond connectivity but different spatial arrangement are called stereoisomers.
Isomerism arising due to different spatial arrangement around a chiral carbon atom is called optical isomerism.
Two non-superimposable mirror image structures are called enantiomers.
A substance which rotates plane polarized light to the right (clockwise) is called dextrorotatory compound.
Halides in which halogen is attached to an sp³ carbon adjacent to a double bond are called allylic halides.
Halides in which halogen is attached to an sp³ carbon next to an aromatic ring are called benzylic halides.
Halides in which halogen is attached to an sp² hybridised carbon of a double bond are called vinylic halides.
The attractive forces between molecules due to temporary dipoles are called van der Waals forces.
A reaction in which one atom or group is replaced by another atom or group is called a substitution reaction.
The reaction of alkyl halide with magnesium in dry ether to form alkyl magnesium halide is called Grignard reaction.
The compound of the type R–Mg–X formed by reaction of alkyl halide with magnesium is called Grignard reagent.
The reaction of alkyl halides with sodium metal in dry ether forming higher alkanes is called Wurtz reaction.
Halogen derivatives in which halogen is directly attached to an aromatic ring are called haloarenes or aryl halides.
Compounds having the same molecular formula but different physical or chemical properties are called isomers.
A substance which rotates plane polarized light to the left (anticlockwise) is called levorotatory compound.
Light vibrating in only one plane perpendicular to the direction of propagation is called plane polarized light.
A substitution reaction in which a nucleophile replaces a leaving group is called a nucleophilic substitution reaction.
A reaction in which the rate depends on the concentration of two reacting species is called a bimolecular reaction.
The inversion of configuration observed in SN₂ reaction due to backside attack is called Walden inversion.
Formation of equal amounts of two enantiomers in an SN₁ reaction is called racemization.
A chemical species that donates an electron pair to form a bond with carbon is called nucleophile.
A solvent capable of hydrogen bonding and stabilizing ions is called polar protic solvent.
A solvent that cannot donate hydrogen for hydrogen bonding and does not strongly solvate anions is called polar aprotic solvent.
Removal of hydrogen halide (HX) from an alkyl halide to form alkene is called dehydrohalogenation reaction.
The rule stating that in elimination reaction the major product is the more substituted alkene is called Saytzeff (Zaitsev) rule.
Key Points
1. On the Basis of Hydrocarbon Skeleton
| Type | General Formula | Description |
|---|---|---|
| Haloalkanes | R–X | Halogen attached to an alkane |
| Haloalkenes | R–CH=CHX | Halogen attached to alkene |
| Haloalkynes | R–C≡C–X | Halogen attached to alkyne |
| Haloarenes | Ar–X | Halogen attached to aromatic ring |
2. On the Basis of the Number of Halogen Atoms
| Type | Description |
|---|---|
| Monohalogen | One halogen atom |
| Dihalogen | Two halogen atoms |
| Trihalogen | Three halogen atoms |
| Polyhalogen | More than three halogens |
3. On the Basis of the Nature of the C–X Bond
(A) Compounds containing sp³ C–X bond
| Type | Description | Example |
|---|---|---|
| Alkyl halide | Halogen attached to the sp³ carbon of the alkyl group | R–CH₂–X |
| Allylic halide | Halogen on carbon next to a double bond | CH₂=CH–CH₂–X |
| Benzylic halide | Halogen on carbon next to the benzene ring | C₆H₅–CH₂–X |
(B) Compounds containing a sp² C–X bond
| Type | Description | Example |
|---|---|---|
| Vinylic halide | Halogen directly on the double bond carbon | CH₂=CH–X |
| Aryl halide | Halogen directly on the aromatic ring | Ar–X |
4. Classification of Alkyl Halides
| Type | Structure | Description |
|---|---|---|
| Primary (1°) | R–CH₂–X | Halogen attached to 1 carbon |
| Secondary (2°) | R–CHX–R | Halogen attached to 2 carbons |
| Tertiary (3°) | R₃C–X | Halogen attached to 3 carbons |
Basic rules
- Alkyl halides → haloalkanes (IUPAC)
- Aryl halides → haloarenes
- Longest chain selection
- Numbering for the lowest position
- Substituents named alphabetically
| Formula | Common name | IUPAC name |
|---|---|---|
| CH₃CH₂Br | Ethyl bromide | Bromoethane |
| CH₃CH(Cl)CH₃ | Isopropyl chloride | 2-Chloropropane |
| (CH₃)₂CH–CH₂Br | Isobutyl bromide | 1-Bromo-2-methylpropane |
| (CH₃)₃CBr | Tert-butyl bromide | 2-Bromo-2-methylpropane |
| (CH₃)₃CCH₂Cl | Neopentyl chloride | 1-Chloro-2,2-dimethylpropane |
| CH≡C–Cl | Chloroacetylene | Chloroethyne |
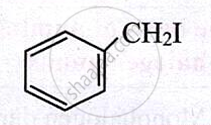 |
Benzyl iodide | Iodophenylmethane |
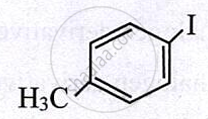 |
p-Iodotoluene | 1-Iodo-4-methylbenzene or 4-Iodotoluene |
- From alkanes
Free radical halogenation:
\[CH_{3}CH_{2}CH_{2}CH_{3}\xrightarrow{Cl_{2}UV-light}CH_{3}CH_{2}CH_{2}CH_{2}Cl+CH_{3}CH_{2}CH(Cl)CH_{3}\] - From alkenes
Addition of hydrogen halide:
C=C + HX → R–X - From alcohols
\[3ROH+PCl_{3}\longrightarrow3R-Cl+H_{3}PO_{3}\]
\[ROH+PCl_{5}\longrightarrow R\longrightarrow Cl+POCl_{3}+HCl\]
\[ROH+HCl\xrightarrow{ZnCl_{2}}R-Cl+H_{2}O\]
\(ROH\xrightarrow{RedP/Cl_{2}}R-Cl\)
\[R—OH+SOCl_2\xrightarrow{\Delta}R—Cl+SO_2\uparrow+HCl\] - Halogen exchange reactions
Finkelstein reaction:
\[R—X+NaI\xrightarrow{Acetone}R—I+NaX\]
Swart's reaction:
\[RBr+AgF\longrightarrow R—F+AgBr\] - From aromatic amines
Sandmeyer reaction:
\[\mathrm{C_{6}H_{5}NH_{2}}\xrightarrow{\mathrm{NaNO_{2}+HX}}\mathrm{C_{6}H_{5}N_{2}^{+}\bar{X}}\xrightarrow{\mathrm{Cu_{2}X_{2}}}\mathrm{C_{6}H_{5}X+N_{2}}\]
- Alkyl halides are colourless when pure but develop colour on exposure to light; they have a sweet smell
- Polarity: Moderately polar
- Solubility: Insoluble in water, soluble in organic solvents
Bond Length
- Increases in order: R–F < R–Cl < R–Br < R–I
Bond Strength
- Decreases in order: R–F > R–Cl > R–Br > R–I
Boiling Point
- Order: R–I > R–Br > R–Cl > R–F
- Increases with an increase in molecular mass
- Decreases with branching
Dipole Moment
-
Decreases as the electronegativity of halogen decreases
Isomeric Dihalobenzenes
- Boiling point: nearly the same
- Melting point: para > ortho/meta
- Optical activity: Ability of a compound to rotate plane-polarised light
- Dextrorotatory (+): rotates light to the right
- Laevorotatory (–): rotates light to the left
- (+) and (–) forms are called optical isomers, and the phenomenon is optical isomerism
- Number of optical isomers = 2ⁿ
(n = number of asymmetric carbon atoms) - Asymmetric carbon/stereocentre: Carbon attached to four different groups
- Chirality: Property of a molecule whose mirror image is non-superimposable
- Enantiomers: Stereoisomers which are non-superimposable mirror images of each other
- Retention: Preservation of spatial arrangement around the asymmetric carbon during reaction
- Inversion of configuration: In the SN2 reaction, backside attack gives a product with the opposite configuration. This complete inversion is called Walden inversion
- Racemic mixture: Equimolar mixture of dextro and laevo isomers. It is optically inactive due to external compensation. This phenomenon is called racemisation.
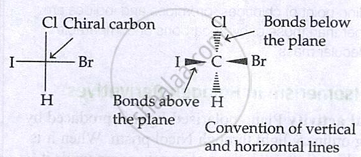
Representation of configuration:
Fischer projection:
- Horizontal bonds → above plane
- Vertical bonds → below plane
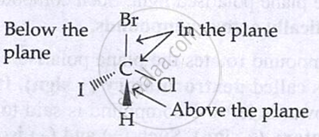
Wedge formula:
- Solid wedge → above plane
- Dashed wedge → below plane
- Straight line → in plane
Alkyl halides mainly undergo substitution and elimination reactions.
1. Nucleophilic Substitution Reactions
SN1 (Unimolecular Nucleophilic Substitution):
- Occurs in two steps:
- Formation of carbocation
- Attack by nucleophile
- First-order reaction (depends only on alkyl halide)
- Reactivity order:
Benzylic > 3° > 2° > 1° > methyl - Leads to racemisation
SN2 (Bimolecular Nucleophilic Substitution):
- Occurs in one step (concerted mechanism)
- Second-order reaction (depends on alkyl halide and nucleophile)
- Reactivity order:
Methyl > 1° > 2° > 3° - Strong nucleophiles increase the rate
- Leads to inversion of configuration
2. Reactions of Alkyl Halides
- With aqueous KOH → Alcohol
- With alcoholic KOH → Alkene (elimination)
- With NaOR → Ether
- With NH₃ → Amine
- With KCN → Nitrile
- With AgCN → Isocyanide
- With KNO₂ → Alkyl nitrite
- With AgNO₂ → Nitro compound
- With Mg (dry ether) → Grignard reagent
- With Na (dry ether) → Alkane (Wurtz reaction)
3. Aryl Halides
- Undergo electrophilic substitution
- Nitration
- Halogenation
- Sulphonation
- Friedel–Crafts reactions
- Show ortho & para directing effect
Common Names:
- Alkyl halide or aryl halide
- e.g., CH₃Cl → Methyl chloride; CH₂=CHCl → Vinyl chloride
IUPAC Names:
- Haloalkane or arylhalide
- Rule 1: Find the longest carbon chain containing the halogen. If a double/triple bond is present, give it priority.
- Rule 2: Number from the end nearer the first substituent. Assign each substituent a position number.
- Multiple same halogens → di-, tri-, tetra- prefix.
- Different halogens → list alphabetically and number to give the alphabetically first halogen the lowest possible number.
Examples:
- CH₃Cl → Chloromethane
- CH₂=CHCl → Chloroethene
- (CH₃)₃CCl → 2-Chloro-2-methylpropane (common: tert-butyl chloride)
- 2-Chloro-1-methylbenzene → o-Chlorotoluene → IUPAC: 1-Chloro-2-methylbenzene
β-Elimination Reaction:
- When alkyl halides are heated with alcoholic KOH or KNH₂, they undergo β-elimination of HX to form an alkene (new π bond).
- The carbon directly attached to X = α-carbon; the carbon adjacent to it = β-carbon.
- Order of reactivity in elimination: R–Cl < R–Br < R–I
Saytzeff's Rule (Zaitsev's Rule):
- In unsymmetrical alkyl halides, hydrogen is preferentially eliminated from the β-carbon with fewer hydrogen atoms → forms the more highly substituted alkene (major product).
- e.g., 2-bromopentane → pent-2-ene (81%) [major] + pent-1-ene (19%) [minor]
Types of Elimination:
- α-elimination: Atom or group lost from the same carbon (gives carbene intermediates).
- β-elimination: H from β-carbon, X from α-carbon → alkene.
- E₁ reaction: Two steps (similar mechanism to SN1)
- E₂ reaction: One step (concerted, anti-periplanar geometry required — similar to SN2 but gives alkene)
Dehydrohalogenation:
- Loss of HX from alkyl halide with alc. KOH → alkene.
\[\ce{\underset{Alky halide}{C_{n}H_{2n + 1}X} ->[Alcholic KOH] \underset{Alkene}{C_{n}H_{2n}} + KX + H2O}\]
- With NaOH, Con. NH₃, t-BuONa, KNH₂, NaNH₂: elimination also occurs.
- Optical isomerism arises due to different spatial arrangement of atoms around a chiral carbon atom, even though the molecular formula and bonding are the same.
- A carbon atom attached to four different atoms or groups makes the molecule chiral and capable of showing optical activity.
- Chiral molecules are not superimposable on their mirror images, just like left and right hands.
- The two mirror-image forms of a chiral compound are called enantiomers and they differ only in the direction in which they rotate plane polarized light.
- Optical activity is the property by which certain compounds rotate the plane of plane polarized light passing through them.
- If the plane of polarized light rotates to the right, the compound is dextrorotatory (+), and if it rotates to the left, it is levorotatory (−).
- Enantiomers have identical physical properties such as melting point and boiling point, but they differ in the direction of optical rotation and in their behavior towards other chiral substances.
- An equimolar mixture of two enantiomers does not rotate plane polarized light because the rotations cancel each other, and such a mixture is optically inactive.
- Dichloromethane (CH₂Cl₂) is a volatile solvent used in aerosols and industrial processes, but overexposure can cause dizziness, nausea, weakness and serious eye damage.
- Chloroform (CHCl₃) is used as a solvent and in the preparation of refrigerants, but prolonged exposure depresses the central nervous system and may damage the liver.
- Carbon tetrachloride (CCl₄) is used as a solvent and cleaning agent, but it is highly toxic to the liver and can affect the central nervous system; it is also suspected to be carcinogenic.
- Iodoform (CHI₃) is a yellow crystalline compound formerly used as an antiseptic, but it can cause irritation to skin, eyes and respiratory system.
- Freons (chlorofluorocarbons, CFCs) are widely used as refrigerants and propellants, but they are responsible for depletion of the ozone layer in the atmosphere.
- Ultraviolet radiation breaks CFC molecules to release chlorine atoms, and a single chlorine atom can destroy thousands of ozone molecules, leading to thinning of the ozone layer.
- DDT is an effective insecticide used to control malaria and typhus, but it is a persistent pollutant that accumulates in fatty tissues, harms wildlife and has serious long-term environmental and health effects.
Important Questions [29]
- Among the following vinylic halide is ______.
- The allylic halide, among the following, is ______.
- Explain optical isomerism in 2-chlorobutane.
- Explain optical activity of 2-chlorobutane.
- Complete the reaction: CHA3CHA2Cl→alc⋅ΔAgCN ?
- Distinguish between SN1 and SN2 mechanism of substitution reaction.
- Write the product formed when alkyl halide reacts with silver nitrite.
- Explain the dehydrohalogenation reaction of 2-chlorobutane.
- What is the action of following on ethyl bromide? alcoholic sodium hydroxide
- What is the action of following on ethyl bromide: Na in dry ether
- Write chemical reactions for the following conversions: Bromobenzene to toluene.
- Write the chemical reaction for the following: Chlorobenzene is heated with fuming H2SO4.
- Write the product formed by chlorobenzene treated with a fuming H2SO4 reaction.
- What is the action of following on ethyl bromide? silver nitrite
- Write the structure of the product formed when chlorobenzene is treated with sodium metal in the presence of dry ether.
- What is the action of following on ethyl bromide? Mg in dry ether
- Write the environmental effects of DDT.
- Draw structure of DDT.
- How is Nitromethane Prepared from the Following? α-nitroalkene
- Complete and Rewrite the Balanced Chemical Equations Chlorobenzene
- How Are the Following Conversions Carried Out? 2-methylbutan-1-ol into 2 -methylbutanoic Acid.
- But-1-ene on Reaction with Hcl in the Presence of Sodium Peroxide Yields
- Iupac Name of Is C6h5 − Ch2 − C − Ch2 − Ch2 − Ch2 − Ch3
- How is Nitromethane Prepared from the Following? Alkyl Halide
- What Are Racemates?
- Draw a Neat, Labelled Energy Profile Diagram for Sn1 Reaction Mechanism.
- Identify ‘A’ and ‘B’ in the Following Reaction
- State and Explain Markownikoff'S Rule with Suitable Example
- Identify the Product ‘D’ in the Following Sequence of Reactions
Concepts [11]
- Classification of Halogen Derivatives
- Nomenclature of Halogen Derivatives
- Methods of Preparation of Alkyl Halides
- Physical Properties of Halogen Derivatives
- Optical Isomerism in Halogen Derivatives
- Chemical Properties of Halogen Derivatives
- Reaction with Active Metals
- Polyhalogen Compounds
- Nomenclature of Haloalkanes and Haloarenes
- Reactions of Haloalkanes - Elimination Reactions
- Overview of Halogen Derivatives
