Advertisements
Advertisements
Question
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Advertisements
Solution
| Factor | SN1 | SN2 |
| Kinetics | 1st order | 2nd order |
| Molecularity | Unimolecular | Bimolecular |
| Number of steps | Two steps | One step |
| Bond making and bond breaking | First the bond in the reactant breaks and then a new bond in the product is formed. | Simultaneous |
| Transition state | Two steps, two transition states | One step, one transition state |
| Direction of attack of nucleophile | Backside attack and frontside attack | Only back side attack |
| Stereochemistry | Racemisation (If the substrate is optically active) | Inversion of configuration (If the substrate is optically active) |
| Type of substrate | Mainly 3° substrates | Mainly 1° substrate |
| Polarity of solvent | Polar protic solvent favorable | Aprotic (non-polar) or solvent with low polarity favourable |
| Intermediate | Intermediate involved | No intermediate |
APPEARS IN
RELATED QUESTIONS
from the following pair would undergo SN2 faster from the other?
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
Benzyl alcohol to benzyl cyanide
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Explain the factors affecting SN1 and SN2 mechanism.
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
How many metameric ethers are represented by the molecular formula C4H10O?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
The compound that reacts the fastest with sodium methoxide is ______.
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the question given below:
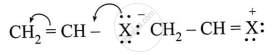
Can react by SN1 mechanism? Justify your answer.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
What is the action of following on ethyl bromide?
alcoholic sodium hydroxide
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Which of the following pair of compounds on heating gives butanenitrile?
Which among the following is NOT benzylic halide?
