Advertisements
Advertisements
प्रश्न
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Advertisements
उत्तर
| Factor | SN1 | SN2 |
| Kinetics | 1st order | 2nd order |
| Molecularity | Unimolecular | Bimolecular |
| Number of steps | Two steps | One step |
| Bond making and bond breaking | First the bond in the reactant breaks and then a new bond in the product is formed. | Simultaneous |
| Transition state | Two steps, two transition states | One step, one transition state |
| Direction of attack of nucleophile | Backside attack and frontside attack | Only back side attack |
| Stereochemistry | Racemisation (If the substrate is optically active) | Inversion of configuration (If the substrate is optically active) |
| Type of substrate | Mainly 3° substrates | Mainly 1° substrate |
| Polarity of solvent | Polar protic solvent favorable | Aprotic (non-polar) or solvent with low polarity favourable |
| Intermediate | Intermediate involved | No intermediate |
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Name the reagent used to bring about the following conversion.
Bromoethane to ethoxyethane
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Convert the following:
Propene to propan-1-ol
The following will react faster by SN1 mechanism
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
The least reactive towards nucleophilic addition reactions is ____________.
Which of the following is least reactive towards SN1 reactions?
Which of the following is least reactive towards nucleophilic addition?
Dehydrohalogenation of an alkyl halide is ____________.
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
Which one is most reactive towards nucleophilic addition reaction?
Which of the following carbocation is the most stable?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the questions given below:
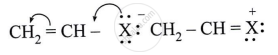
Name the type of halogen derivative.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
Identify the substrate 'X' in the following reaction.
\[\ce{\underset{}{X + O2} ->[i) Co-naphtenate 423 K][ii) dil HClΔ] \underset{\underset{}{}}{Phenol + Acetone}}\]
