Advertisements
Advertisements
प्रश्न
Convert the following:
Ethanol to propane nitrile
Advertisements
उत्तर
\[\ce{\underset{Ethanol}{CH3CH2OH} + HCl ->[Groove's][process]\underset{Chloroethane}{CH3CH2Cl} ->[Alc.KCN,\Delta][-H2O] \underset{Propane nitrile}{CH3CH2CN}}\]
APPEARS IN
संबंधित प्रश्न
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
2-Chloropropane to propan-1-ol
Propane nitrile can be prepared by heating ____________
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is a primary halide?
The least reactive towards nucleophilic addition reactions is ____________.
Which of the following is least reactive towards nucleophilic addition?
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
Which among the following statements is not correct about SN2 reaction mechanism?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the questions given below:
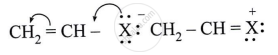
Name the type of halogen derivative.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Identify the substrate 'X' in the following reaction.
\[\ce{\underset{}{X + O2} ->[i) Co-naphtenate 423 K][ii) dil HClΔ] \underset{\underset{}{}}{Phenol + Acetone}}\]
Which among the following is NOT benzylic halide?
