Advertisements
Advertisements
प्रश्न
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Advertisements
उत्तर
\[\ce{\underset{(Ethyl chloride)}{CH3CH2Cl} ->[AgCN][alc.\Delta] \underset{(Ethyl isocyanide)}{CH3 - CH2 - NC}}\]
APPEARS IN
संबंधित प्रश्न
Complete the following reaction giving major products.
\[\begin{array}{cc}\ce{CH3 - CH = CH3 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{.........................}\\
\ce{OH\phantom{.......................}}\end{array}\]
Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Name the type of halogen derivative.
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain the factors affecting SN1 and SN2 mechanism.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
Dehydrohalogenation of an alkyl halide is ____________.
The following mechanism has been proposed for the reaction of NO2 with F2 to form NO2F:
\[\ce{NO2_{(g)} + F2_{(g)} -> NO2F_{(g)} + F_{(g)} (slow)}\]
\[\ce{F_{(g)} + NO2_{(g)} -> NO2F_{(g)} (fast)}\]
The order of the reaction with respect to NO2(g)
How many metameric ethers are represented by the molecular formula C4H10O?
Identify major product 'B' in the following reaction.
\[\ce{But-1-ene ->[HBr][Peroxide] A ->[AgCN][\Delta] B}\]
Write the product formed when alkyl halide reacts with silver nitrite.
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
The order of reactivities of the following alkyl halides for an SN2 reaction is ______.
The order of reactivity of the given haloalkanes towards nucleophiles is:
Which among the following statements is not correct about SN2 reaction mechanism?
With which halogen the reactions, of alkanes are explosive?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Complete the following reaction giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the question given below:
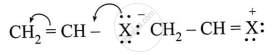
Can react by SN1 mechanism? Justify your answer.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Which among the following is NOT benzylic halide?
