Advertisements
Advertisements
प्रश्न
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Advertisements
उत्तर
\phantom{.}\ce{Br}\phantom{................................................................}\ce{Br}\phantom{.............................................}\\
\phantom{...}|\phantom{..................................................................}|\phantom{................................................}\\
\ce{\underset{\text{2-Bromobutane}}{H3C - CH - CH2CH3}->[Alc.KOH][-KBr]\underset{\underset{(Major) (A)}{But-2-ene}}{H3C - CH = CH - CH3}->[Br2] H3C - CH - CH - CH3 ->[NaNH2][Double dehydrohalogen reaction] \underset{\text{But-2-yne (C)}}{H3C - C ≡ C-CH3}}\\
\phantom{.........}|\\
\phantom{................}\ce{\underset{\text{2,3-Dibromobutane (B)}}{Br}}
\end{array}\]
संबंधित प्रश्न
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Complete the following reaction giving major products.

Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Convert the following:
Propene to propan-1-ol
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
Which of the following is a primary halide?
The least reactive towards nucleophilic addition reactions is ____________.
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
How many metameric ethers are represented by the molecular formula C4H10O?
Which one is most reactive towards nucleophilic addition reaction?
Which of the following carbocation is the most stable?
Write the product formed when alkyl halide reacts with silver nitrite.
Observe the following and answer the question given below:
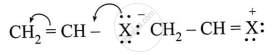
Can react by SN1 mechanism? Justify your answer.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction, giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
Which of the following pair of compounds on heating gives butanenitrile?
