Advertisements
Advertisements
प्रश्न
Convert the following:
tert-Butyl bromide to isobutyl bromide
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{\phantom{.........}Br\phantom{.........................................................}H\phantom{............}}\\
\phantom{........}|\phantom{...........................................................}|\phantom{...........}\\
\ce{H3C - C - CH3 +\underset{(Alc.)}{KOH} -> H3C - C = CH2 + HBr ->[Peroxide] H3C - C - CH2Br}\\
\phantom{...}|\phantom{............................}|\phantom{...............................}|\phantom{.....}\\
\ce{\underset{\underset{(2-Bromo-2-methyl propane)}{Tert-Butyl bromide}}{CH3}\phantom{...............}\underset{\underset{(2–Methylprop-1-ene)}{Isobutene}}{CH3}\phantom{....................}\underset{Isobutyl bromide}{CH3}\phantom{...}}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Convert the following:
Propene to propan-1-ol
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is least reactive towards SN1 reactions?
Which one is most reactive towards nucleophilic addition reaction?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
With which halogen the reactions, of alkanes are explosive?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the question given below:
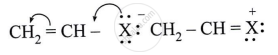
Comment on the bond length of C-X bond in it.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Identify the substrate 'X' in the following reaction.
\[\ce{\underset{}{X + O2} ->[i) Co-naphtenate 423 K][ii) dil HClΔ] \underset{\underset{}{}}{Phenol + Acetone}}\]
Which of the following reagent is used for conversion of alkyl halide into nitrile?
