Advertisements
Advertisements
प्रश्न
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Advertisements
उत्तर
- The low reactivity of aryl halides is due to the resonance effect and sp2 hybrid state of carbon to which halogen atom is attached.
- In aryl halides, one of the lone pairs of electrons on the halogen atom is in conjugation with π-electrons of the ring. Due to resonance, the C–X bond acquires partial double bond character. Thus, the C–X bond in aryl halides is stronger and shorter than alkyl halides. Hence, it is difficult to break C–X bond in aryl halides.
- Further, the phenyl cation produced due to the self-ionization of aryl halide will not be stabilised by resonance. This rules out the possibility of SN1 mechanism. Also, the backside attack of nucleophiles is blocked by the aromatic ring. This rules out the possibility of SN2 mechanism. As a result, nucleophilic substitution reaction involving cleavage of C–X bond in haloarenes proceeds with difficulty.
- Therefore, aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
संबंधित प्रश्न
Complete the following reaction giving major products.

Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Convert the following:
Ethanol to propane nitrile
Convert the following:
2-Chloropropane to propan-1-ol
Observe the following and answer the question given below.

Name the type of halogen derivative.
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
Which one is most reactive towards nucleophilic addition reaction?
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following carbocation is the most stable?
Which of the following is FALSE regarding SN2 reaction mechanism?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
Identify major product 'B' in the following reaction.
\[\ce{But-1-ene ->[HBr][Peroxide] A ->[AgCN][\Delta] B}\]
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
Which among the following statements is not correct about SN2 reaction mechanism?
Observe the following and answer the questions given below:
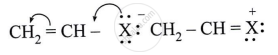
Name the type of halogen derivative.
Observe the following and answer the question given below:
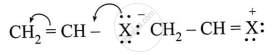
Can react by SN1 mechanism? Justify your answer.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Which of the following reagent is used for conversion of alkyl halide into nitrile?
