Advertisements
Advertisements
Question
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
Advertisements
Solution

APPEARS IN
RELATED QUESTIONS
Complete the following reaction giving major products.
\[\begin{array}{cc}\ce{CH3 - CH = CH3 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{.........................}\\
\ce{OH\phantom{.......................}}\end{array}\]
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
tert-Butyl bromide to isobutyl bromide
The following will react faster by SN1 mechanism
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
The least reactive towards nucleophilic addition reactions is ____________.
Which of the following is least reactive towards nucleophilic addition?
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following carbocation is the most stable?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
Which among the following statements is not correct about SN2 reaction mechanism?
With which halogen the reactions, of alkanes are explosive?
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the questions given below:
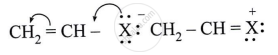
Name the type of halogen derivative.
Observe the following and answer the question given below:
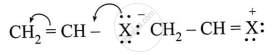
Can react by SN1 mechanism? Justify your answer.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Which among the following is NOT benzylic halide?
Which of the following reagent is used for conversion of alkyl halide into nitrile?
