Advertisements
Advertisements
Question
The following will react faster by SN1 mechanism
Options
1-chloropropane
2-chloropropane
2-chloro-2-methylpropane
chloroethane
Advertisements
Solution
2-chloro-2-methylpropane
Explanation:
Reactivity order of alkyl halides towards SN1 mechanism: 3° > 2° > 1° > CH3X
RELATED QUESTIONS
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Name the reagent used to bring about the following conversion.
Bromoethane to ethoxyethane
Name the reagent used to bring about the following conversion.
1-Chloropropane to 1-nitropropane
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Observe the following and answer the question given below.

Name the type of halogen derivative.
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is a primary halide?
The least reactive towards nucleophilic addition reactions is ____________.
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
Identify major product 'B' in the following reaction.
\[\ce{But-1-ene ->[HBr][Peroxide] A ->[AgCN][\Delta] B}\]
Write the product formed when alkyl halide reacts with silver nitrite.
The compound that reacts the fastest with sodium methoxide is ______.
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Observe the following and answer the question given below:
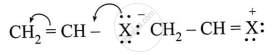
Can react by SN1 mechanism? Justify your answer.
Discuss β - elimination reaction.
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
Which of the following pair of compounds on heating gives butanenitrile?
