Advertisements
Advertisements
Question
Complete the following reaction giving major products.
\[\begin{array}{cc}\ce{CH3 - CH = CH3 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{.........................}\\
\ce{OH\phantom{.......................}}\end{array}\]
Advertisements
Solution
\[\begin{array}{cc}
\ce{3CH3-CH-CH3->[Red P/Br2][(Bromination)]3CH3-CH-CH3 +H3PO3->[Ag2O/H2O]3CH3-CH-CH3}\\
\phantom{}|\phantom{.............................}|\phantom{.....................................}|\phantom{}\\
\ce{\underset{2-Hydroxypropane}{OH}\phantom{................}\underset{2-Bromopropane(A)}{Br}\phantom{.........................}\underset{Propan-2-ol(B)}{OH}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Name the reagent used to bring about the following conversion.
Bromoethane to ethoxyethane
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is a primary halide?
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
Which of the following is FALSE regarding SN2 reaction mechanism?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
The order of reactivities of the following alkyl halides for an SN2 reaction is ______.
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
The order of reactivity of the given haloalkanes towards nucleophiles is:
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Observe the following and answer the question given below:
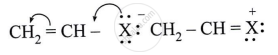
Comment on the bond length of C-X bond in it.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Define and explain the SN1 mechanism with a suitable example.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Which among the following is NOT benzylic halide?
