Advertisements
Advertisements
Question
How is nitromethane prepared from the following?
alkyl halide
Advertisements
Solution
Preparation of nitromethane from alkyl halide:
Bromomethane reacts with silver nitrite (AgNO2), to give nitromethane

Nitromethane is obtained in good yield by action of sodium or potassium nitrite on
bromomethane, in presence of solvent dimethyl sulphoxide or N,N-dimethylformamide

APPEARS IN
RELATED QUESTIONS
Write the structure of the following compound:
1-Chloro-4-ethylcyclohexane
Write the structure of the following compound:
1,4-Dibromobut-2-ene
Write the structure of the following compound:
1-Bromo-4-sec. butyl-2-methylbenzene
Write structures of different dihalogen derivatives of propane.
Give the IUPAC name of the following compound:
CH3CH(Cl)CH(Br)CH3
Give the IUPAC name of the following compound:
CHF2CBrClF
Give the IUPAC name of the following compound:
(CCl3)3CCl
Write the structure of the following organic halogen compound.
p-Bromochlorobenzene
But-1-ene on reaction with HCl in the presence of sodium peroxide yields _______.
Write the structure of 1-Bromo-4-chlorobut-2-ene
Write the mechanism (using curved arrow notation) of the following reaction:

Write the product(s) in the following reaction:

Complete and rewrite the balanced chemical equations

Bromomethane, Chloromethane, Dibromomethane, 1-Chloropropane, Isopropyl chloride, 1-Chlorobutane all are:
Among the following which one can have a meso form?
Ethylene dichloride can be prepared by adding HCl to ____________.
Benzene diazonium chloride reacts with hypophosphorous acid to produce ____________.
Which of the following is most stable?
\[\ce{CH3CH2CH2Cl ->[alc. KOH] B ->[HBr] C ->[Na/ether] D}\]
In the above reaction, the product D is:
Which of the following compounds are gem-dihalides?
(i) Ethylidene chloride
(ii) Ethylene dichloride
(iii) Methylene chloride
(iv) Benzyl chloride
Arrange the following compounds in order of decreasing acidity:
| (I) |  |
| (II) | 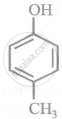 |
| (III) | 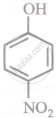 |
| (IV) |  |
Write the structure of the following compound:
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert. Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Give the IUPAC name of the following compound:
ClCH2C ≡ CCH2Br
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structures of the following organic halogen compounds.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Give the IUPAC name of the following compound:
CICH2C ≡ CCH2Br
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following compound:
4-tert-Butyl-3-iodoheptane
Give the IUPAC names of the following compound:
\[\ce{ClCH2C ≡ CCH2Br}\]
Write the structure of the following compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following compound:
1,4-Dibromobut-2-ene
Give the IUPAC name of the following compound:
\[\ce{ClCH2C ≡ CCH2Br}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound.
4 – tert – Butyl -3 -iodoheptane
Write structure of the following compound:
1,4-Dibromobut-2-ene
Write structure of the following compound:
1,4-Dibromobut-2-ene
Write structure of the following compound:
4-tert. Butyl-3-iodoheptane
Write structure of the following compound:
4-tert. Butyl-3-iodoheptane
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound:
1,4-Dibromobut 2 ene
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
