Advertisements
Advertisements
Question
Write structures of different dihalogen derivatives of propane.
Advertisements
Solution 1
Four isomeric dihalogen derivatives of propane (CH3 CH2 CH3) are possible.
\[\ce{\underset{1, 3-dibromopropane}{BrCH2 - CH2 - CH2Br}}\]
\[\begin{array}{cc}
\ce{BrCH2 - CH - CH3}\\
|\\
\phantom{.}\ce{\underset{1, 2-dibromopropane}{Br}}
\end{array}\]
\[\begin{array}{cc}
\ce{Br - CH - CH2 - CH3}\\
|\phantom{...........}\\
\ce{\underset{1, 1-dibromopropane}{Br}}\phantom{..........}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3 - C - CH3}\\
/\phantom{.}\backslash\\
\phantom{..}\ce{\underset{2, 2-dibromopropane}{Br\phantom{..}Br}}
\end{array}\]
Solution 2
The structures of all possible dihalogen derivatives of propane are:
\[\ce{\underset{1, 3-dihalopropane}{X - CH2 - CH2 - CH2 - X}}\]
\[\begin{array}{cc}
\ce{X}\phantom{.......}\\
|\phantom{.......}\\
\ce{\underset{1, 2-dihalopropane}{H3C - CH - CH2 - X}}\
\end{array}\]
\[\begin{array}{cc}
\ce{X}\\
|\\
\ce{H3C - C - CH2}\\
|\\
\ce{\underset{2, 2-dihalopropane}{X}}\\
\end{array}\]
\[\begin{array}{cc}
\phantom{.............}\ce{X}\\
\phantom{.............}|\\
\ce{H3C - CH2 - CH}\\
\phantom{.............}|\\
\phantom{.............}\ce{\underset{1, 1-dihalopropane}{X}}\\
\end{array}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
RELATED QUESTIONS
Write the structure of the following compound:
1,4-Dibromobut-2-ene
Give the IUPAC name of the following compound:
(CH3)3CCH=CClC6H4I-p
Write the structure of the following organic halogen compound.
p-Bromochlorobenzene
But-1-ene on reaction with HCl in the presence of sodium peroxide yields _______.
Write the product(s) in the following reactions:

Write the structures of the main products in the following reactions :

Among the following which one can have a meso form?
Which of the following will have a mesoisomer also?
\[\begin{array}{cc}
\ce{CH3-CH-CH=CH2 + HBr -> A}\\
|\phantom{.....................}\\
\ce{CH3}\phantom{..................}
\end{array}\]
'A' is:
Arrange the following compounds in order of decreasing acidity:
| (I) |  |
| (II) | 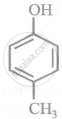 |
| (III) | 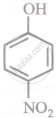 |
| (IV) |  |
Write the structure of the following organic halogen compound.
4-tert. Butyl-3-iodoheptane
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structures of the following organic halogen compound
1,4-Dibromobut-2-ene
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound.
4 – tert – Butyl -3 -iodoheptane
Write structure of the following compound:
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound:
1,4-Dibromobut 2 ene
