Advertisements
Advertisements
Question
The allylic halide, among the following, is ______.
Options
\[\begin{array}{cc}
\phantom{}\ce{R - CH - R}\phantom{}\\
|\phantom{..}\\
\ce{X}\phantom{..}
\end{array}\]CH2 = CH – X
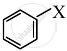
CH2 = CH – CH2 – X
Advertisements
Solution
The allylic halide, among the following is CH2 = CH – CH2 – X.
Explanation:
In allylic halides, halogen atom is bonded to a sp3 hybridized carbon atom next to a carbon-carbon double bond.
| CH2 = CH – CH2 – X | 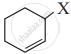 |
APPEARS IN
RELATED QUESTIONS
The correct order of increasing reactivity of C-X bond towards nucleophile in the following compounds is:
(I)
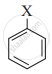
(II)
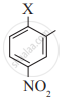
(III) (CH3)3 C-X
(IV) (CH3)2 CH-X
\[\ce{CH3 - CH = CH2 ->[HI][Peroxide]}\]
The major product of the above reaction is,
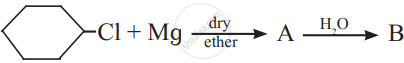
The product ‘B’ in the above reaction sequence is,
Choose the most correct option.
Which of the following is likely to undergo racemization during alkaline hydrolysis?
(I)
\[\begin{array}{cc}
\ce{CH3-CH-C2H5}\\
|\\
\ce{Cl}\end{array}\]
(II)
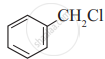
(III)
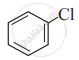
(IV)
\[\begin{array}{cc}
\ce{\phantom{.......}CH3}\\
\phantom{.....}|\\
\ce{CH3-CH}\\
\phantom{.....}|\\
\ce{\phantom{..........}CH2Cl}
\end{array}\]
The common name of 1-chloro-2, 2-dimethyl propane is ______.
The bond line formula of 1-iodo-2, 3-dimethyl pentane is
When propene reacts with HCl in presence of peroxide, the product is ________.
The conversion of 2-methylpropan-1-ol to 2-methylpropan-2-ol is ______.
Alkyl chloride containing 4° carbon atom(s) would be ____________.
Which of the following compounds is obtained when t-butyl bromide is treated with alcoholic ammonia?
The number of possible monohalogen derivatives for the alkyl halide having molecular formula C4H9X is ____________.
Identify 'B' in the following series of reactions.
\[\ce{Ethanol ->[NaBr][H2SO4, \Delta] A ->[Mg][Dry ether] B}\]
What is the IUPAC name of neopentyl chloride?
Identify A and B respectively in the following conversion.
\[\ce{Ethene ->[A] Bromoethane ->[B][\Delta] Ethyl propionate}\]
The IUPAC name of isobutyl bromide is ____________.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry either] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Identify the chiral molecule from the following.
Identify the chiral molecule from the following.
Identify A and B:
\[\ce{C6H5CH2Br ->[Alc.][KCN] A ->[Na/Ethanol] B}\]
Identify the chiral molecule from the following.
Among the following vinylic halide is ______.
