Advertisements
Advertisements
Question
Write the mechanism of hydration of ethene to yield ethanol.
Advertisements
Solution
The mechanism of hydration of ethene to yield ethanol involves the following three steps:
Step 1: Protonation of alkene to form carbocation by electrophilic attack of H3O+.
\[\ce{H2O + H+ -> H3O+}\]
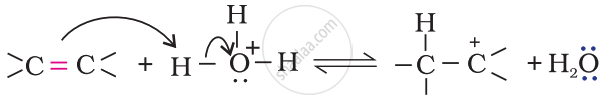
Step 2: Nucleophilic attack of water on carbocation.
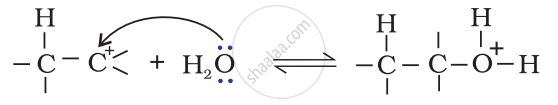
Step 3: Deprotonation to form an alcohol.

APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]

How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Aldehydes react with Grignard reagent to produce ____________.
Ketones react with Grignard reagent to produce ____________.
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
The reagent used for dehydration of an alcohol is
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
When glycol is heated with dicorboxylic acid the product are
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
