Advertisements
Advertisements
Question
State Charles's law.
Advertisements
Solution
Charles's Law
At constant pressure, the volume of a given mass of a dry gas increases or decreases by 1/273rd of its original volume at 0°C for each degree centigrade rise or fall in temperature.
V ∝ T (at constant pressure)
At temperature T1 (K) and volume V1 (cm3):
`"V"_1 ∝ "T"_1 "or" "V"_1/"T"_1 = "K"` = constant...(i)
At temperature T2 (K) and volume V2 (cm3):
`"V"_2 ∝ "T"_2 "or" "V"_2/"T"_2 = "K"` = constant….(ii)
From (i) and (ii),
`"V"_1/"T"_1 = "V"_2/"T"_2` = constant
For Temperature = Conversion from Celsius to Kelvin
1 K = °C + 273
Example:
20°C = 20 + 273 = 293 K
APPEARS IN
RELATED QUESTIONS
State the law which is represented by the following graph:
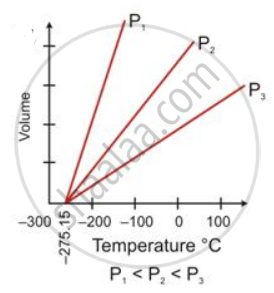
How did Charles's law lead to the concept of an absolute scale of temperature?
Choose the correct answer:
The absolute temperature value that corresponds to 27°C is
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant?
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Fill in the blank with the correct word, from the words in option:
At -273°C the volume of a gas is theoretically ______.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
The following question refers to one mole of chlorine gas.
What volume will it occupy at 273°C?
