Advertisements
Advertisements
Question
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Advertisements
Solution
Initial conditions:
V1 = Initial volume of the gas = V
T1 = Initial temperature of the gas = ?
Final conditions:
V2 (Final volume) = reduce by 20%
= V - `20/100` of V
= `(80"V")/100`
= `(4"V")/5`
T2 = Final temperature of the gas = 157°C = 157 + 273 = 430 K
By Charles Law:
`"V"_1/"T"_1 = "V"_2/"T"_2`
Substituting the values:
`"V"/"T"_1 = ((4"V")/5)/430`
`1/"T"_1 = 4/(5 xx 430)`
T1 = `(430 xx 5)/4`
T1 = 537.5 K
Therefore, final temperature of the gas = 537.5 K - 273 K = 264.5°C
APPEARS IN
RELATED QUESTIONS
Give the assumptions of the kinetic molecular theory.
Explain Charles's law on the basis of the kinetic theory of matter.
State the law which is represented by the following graph:
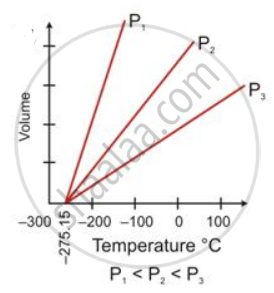
Give reasons for the following:
Mountaineers carry oxygen cylinders with them.
How did Charles's law lead to the concept of an absolute scale of temperature?
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
Correct the following statement:
The volume of a fixed mass of a gas is directly proportional to its temperature, pressure remaining constant.
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
At a given temperature the pressure of a gas reduces to 75% of its initial value and the volume increases by 40% of its initial value. Find this temperature if the initial temperature was -10°C.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
