Advertisements
Advertisements
Question
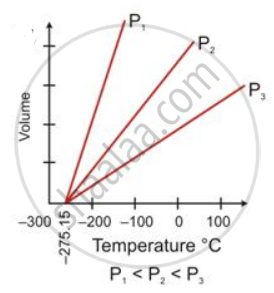
State the law which is represented by the following graph:
Advertisements
Solution
Charles's law
APPEARS IN
RELATED QUESTIONS
Explain Charles's law on the basis of the kinetic theory of matter.
Give reasons for the following:
Mountaineers carry oxygen cylinders with them.
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
An LPG cylinder can withstand a pressure of 14.9 atmospheres. The pressure gauge of the cylinder indicates 12 atmospheres at 27°C. Because of a sudden fire in the building, the temperature rises. At what temperature will the cylinder explode?
Give its
(i) graphical representation,
(ii) mathematical expression
(iii) significance
Correct the following statement:
The volume of a fixed mass of a gas is directly proportional to its temperature, pressure remaining constant.
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
The following question refers to one mole of chlorine gas.
What volume will it occupy at 273°C?
