Advertisements
Advertisements
प्रश्न
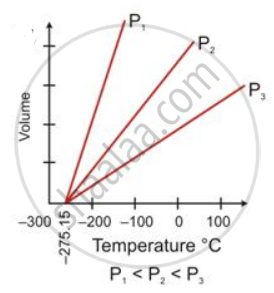
State the law which is represented by the following graph:
Advertisements
उत्तर
Charles's law
APPEARS IN
संबंधित प्रश्न
Give the assumptions of the kinetic molecular theory.
Explain Charles's law on the basis of the kinetic theory of matter.
Give reasons for the following:
Mountaineers carry oxygen cylinders with them.
Choose the correct answer:
The absolute temperature value that corresponds to 27°C is
Calculate the final volume of a gas 'X' if the original pressure of the gas at STP is doubled and its temperature is increased three times.
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was −15°C, find the temperature to which it was heated.
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
Give its
(i) graphical representation,
(ii) mathematical expression
(iii) significance
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
