Advertisements
Advertisements
प्रश्न
Give the assumptions of the kinetic molecular theory.
Advertisements
उत्तर
- Gases are made of tiny particles that move in all possible directions at all possible speeds. The size of the molecules is small as compared to the volume of the occupied gas.
- There is no force of attraction between gas particles or between the particles and the walls of the container. So, the particles are free to move in the entire space available to them.
- The moving particles of gas collide with each other and with the walls of the container. Because of these collisions, gas molecules exert pressure. Gases exert the same pressure in all directions.
- There is a large interparticle space between gas molecules, and this accounts for high compressibility of gases.
- The volume of a gas increases with a decrease in pressure and an increase in temperature.
- Gases have a low density as they have large intermolecular spaces between their molecules.
- Gases have a natural tendency to mix with one and other because of large intermolecular spaces. So, two gases when mixed form a homogeneous gaseous mixture.
- The intermolecular space of a gas is reduced because of cooling. Molecules come closer resulting in liquefaction of the gas.
APPEARS IN
संबंधित प्रश्न
State the law which is represented by the following graph:
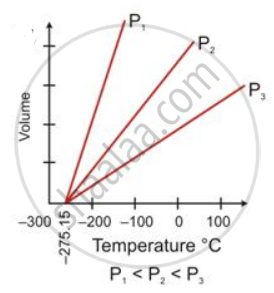
How did Charles's law lead to the concept of an absolute scale of temperature?
State the following:
Ice point in absolute temperature
Choose the correct answer:
The volume-temperature relationship is given by
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
Fill in the blank with the correct word, from the words in option:
At -273°C the volume of a gas is theoretically ______.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
