Advertisements
Advertisements
प्रश्न
State Charles's law.
Advertisements
उत्तर
Charles's Law
At constant pressure, the volume of a given mass of a dry gas increases or decreases by 1/273rd of its original volume at 0°C for each degree centigrade rise or fall in temperature.
V ∝ T (at constant pressure)
At temperature T1 (K) and volume V1 (cm3):
`"V"_1 ∝ "T"_1 "or" "V"_1/"T"_1 = "K"` = constant...(i)
At temperature T2 (K) and volume V2 (cm3):
`"V"_2 ∝ "T"_2 "or" "V"_2/"T"_2 = "K"` = constant….(ii)
From (i) and (ii),
`"V"_1/"T"_1 = "V"_2/"T"_2` = constant
For Temperature = Conversion from Celsius to Kelvin
1 K = °C + 273
Example:
20°C = 20 + 273 = 293 K
APPEARS IN
संबंधित प्रश्न
Explain Charles's law on the basis of the kinetic theory of matter.
State the law which is represented by the following graph:
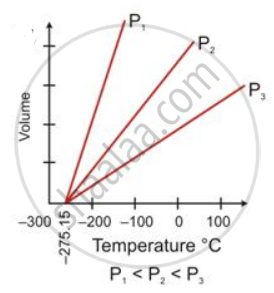
How did Charles's law lead to the concept of an absolute scale of temperature?
State the following:
Ice point in absolute temperature
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
An LPG cylinder can withstand a pressure of 14.9 atmospheres. The pressure gauge of the cylinder indicates 12 atmospheres at 27°C. Because of a sudden fire in the building, the temperature rises. At what temperature will the cylinder explode?
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
At a given temperature the pressure of a gas reduces to 75% of its initial value and the volume increases by 40% of its initial value. Find this temperature if the initial temperature was -10°C.
Fill in the blank with the correct word, from the words in option:
If the temperature of a fixed mass of a gas is kept constant and the pressure is increased, the volume correspondingly _______.
Fill in the blank with the correct word, from the words in option:
At -273°C the volume of a gas is theoretically ______.
