Advertisements
Advertisements
Question
The pressure on one mole of gas at s.t.p. is doubled and the temperature is raised to 546 K. What is the final volume of the gas ? [one mole of a gas occupies a volume of 22.4 litres at stp.]
Advertisements
Solution
One mole of gas occupies a volume of 22.4 lit. at s.t.p.
∴ V1 = 22.4 lit.
P1 = 1 atm.
T1 = 273 K
V2 = ?
P2 = 2 atm.
T2 = 546 K
Using gas equation `("P"_1"V"_1)/"T"_1 = ("P"_2"V"_2)/"T"_2`
V2 = `("P"_1"V"_1"T"_2)/("T"_1"P"_2)`
`= (1xx22.4xx546)/(273xx2)` = 22.4 lits.
APPEARS IN
RELATED QUESTIONS
Explain Charles's law on the basis of the kinetic theory of matter.
State the law which is represented by the following graph:
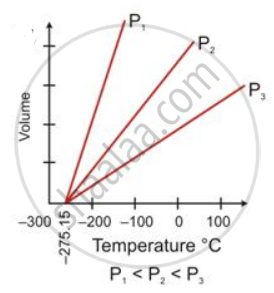
How did Charles's law lead to the concept of an absolute scale of temperature?
State the following:
Ice point in absolute temperature
Choose the correct answer:
The volume-temperature relationship is given by
Taking the volume of hydrogen as calculated in Q.19, what change must be made in Kelvin (absolute) temperature to return the volume to 2500 cm3 (pressure remaining constant)?
State Charles's law.
At a given temperature the pressure of a gas reduces to 75% of its initial value and the volume increases by 40% of its initial value. Find this temperature if the initial temperature was -10°C.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
According to Charles’s law, at constant pressure, the temperature is inversely proportional to volume.
